Although recent guidelines focus on asthma control, a significant number of patients remains poorly controlled.
To evaluate the longitudinal effects of corticosteroids on lung function, inflammation and asthma control.
22 asthmatic children and adolescents (10.5 ± 2.55 years) participated of three visits (2 months a part). On each visit spirometry, fractional exhaled nitric oxide (FeNO), level of asthma control (Asthma control test-ACT), and asthma severity were assessed. The beclomethasone (Clenil® 250 μg/jato-Laboratory Chiesi-Brazil), at a dose of 1 jet 12/12 h, started on the first visit.
One patient was classified as having controlled asthma, 10 partially controlled asthma (PCA), and 11 uncontrolled asthma (UC). On the third visit, 14 patients were classified as PCA and 7 UC. The FEV1/FVC (%) significantly increased from baseline to the third visit (p = 0.0098). The number of asthma patients with low levels of FeNO increased from 9 to 16 patients and the number of asthma patients with intermediate levels of FeNO reduced from 7 to 2 patients, considering the first and third visits. Dispersion of FeNO levels were widely in the study sample. As expected, the FeNO correlated inversely with FEV1/FVC (%) in all evaluations. The baseline ACT score was positively correlated with baseline FEV1 % (r = 0.4480, p = 0.0417). FeNO levels, FEV1 (%), FEV1/CVF (%), and ACT scores did not differ between atopic and non-atopic subgroups in 3 time points.
Treatment with corticosteroids improved the classification of the asthma control level. Despite the great dispersion of the FeNO levels, this biomarker correlated inversely with Tiffeneau index in the three evaluations.
Asthma control test, Atopy, Inflammation, Nitric oxide, IgE sensitization
Asthma is a heterogeneous disease characterized by chronic inflammation of the airways, defined by history of respiratory symptoms of wheezing, breathlessness, chest tightness and coughing, along with chronic airflow limitation [1]. This disease presents different phenotypes [2] which may be related to the natural history of the disease, with immune, inflammatory and structural characteristics of the airways and clinical control. The inflammation, structural changes caused by the maintenance of the inflammatory process and the imbalance between extracellular matrix synthesis and degradation lead to reduction in airway caliber and bronchial hyperresponsiveness [1]. Epidemiological studies have shown that asthma is now the most common chronic disease among children and is a major cause of absenteeism from school and adults at work. Furthermore, asthma is associated with a significant mortality rate [3,4].
Pulmonary function test is useful for evaluating airflow obstruction, reversibility of airway obstruction by bronchodilator test and excessive variation in lung function during the visits. Forced expiratory volume in the first second of forced vital capacity (FEV1) down is a strong independent predictor of future risk of exacerbations and is a risk factor for the decline in lung function [1].
The role of nitric oxide exhaled fraction (FeNO) in the management of asthma is not well established. The exhaled nitric oxide is considered a marker of airway inflammation and is closely associated with bronchial eosinophilia [5]. According to some authors, this biomarker of airway can be used to define the phenotype of asthma, predict the development and exacerbation of the disease, predict responsiveness to corticosteroids and assess the level of asthma control [6,7]. Furthermore, it was demonstrated that patients with higher levels of exhaled nitric oxide have higher rates of decline in FEV1 [8]. However, none of the studies conducted so far has shown that the use of FeNO was better than current asthma guidelines to control asthma exacerbations. Furthermore, there is a wide variation in exhaled nitric oxide levels between individuals, which may reflect the heterogeneity of the activity of nitric oxide synthase (NOS) in the epithelium and/or the contribution of other non-eosinophilic epithelial NOS activity [9].
The goal of asthma management is to control the disease, which refers to the extent to which the manifestations of asthma are reduced or resolved by the treatment, and comprises two distinct domains: symptom control and reducing future risk events adverse [1]. Several instruments have been used to assess asthma control. The asthma control test (ACT), one of the tools most commonly used in clinical practice, consists of five questions that assess signs and symptoms and rescue medication use (β2 agonist short duration) in the last four weeks [10]. The validity and responsiveness of the TCA to changes in asthma control have been documented in several studies [11-14].
Drug treatment is based on the reduction of the inflammatory process, with the inhaled corticosteroid election therapy in asthma. Inhaled corticosteroids reduce the frequency and severity of exacerbations, the number of hospitalizations and visits to emergency services, as well as improved the quality of life, lung function and hyperactivity bronchial as well, decreases exercise-induced bronchoconstriction [15]. Recent study Marchioro, et al., it showed that the vast majority of patients with asthma in Brazil does not have their disease under control, according to international criteria. According to these authors, the maintenance medications of asthma control are still underused in Brazil, and the use of relief medications and oral corticosteroids are more frequent in patients with partially controlled or uncontrolled asthma.
Asthma is typically recognized by an eosinophilic inflammatory airway disease. However, the association between inflammation and clinical control of asthma is not well established. Studies have shown that the reduction of corticosteroids is associated with loss of clinical control and increased eosinophils in sputum. Due to inadequate control of asthma, this study evaluated the effect of corticosteroid on the association between lung function (used for diagnosis and classification of asthma), FeNO (an eosinophilic inflammation marker of the airways) and asthma control (control test asthma) in children and adolescents without prior use of inhaled corticosteroids.
The sample size was obtained to detect a difference of 3/5 of the standard deviation of the difference between two measurements of the same individual, lengthwise. The 5% level of significance and power of 80% was obtained n = 22. The formula [16] was used:
Where and Sd denote the mean and standard deviation of the differences between the two measures (lengthwise) and Z(1-α/2) and Z(1-β) are quantiles of the normal distribution related to the level of significance and power, respectively. The sample size was 22 obtained.
This is a prospective, longitudinal study involving 22 children and adolescents with asthma, classified with persistent asthma and moderate to severe persistent asthma. The study was conducted at the Secondary Reference Unit Campos Sales in Belo Horizonte, Minas Gerais, in the period from February to December 2015. During this period there were three clinical evaluations, with an average interval of two months each. The participants were referred by general practitioners and pediatricians in primary care settings.
After the rest period and fasting for at least 1 hour, were carried out of nitric oxide exhaled and anthropometric measurements. After the clinical examination were performed spirometry and bronchodilator test. It was later performed allergy tests and reading after 15 minutes. Finally, we applied the asthma control test.
The patients underwent skin test Immunotech® (Rio de Janeiro, Rio de Janeiro, Brazil) whose panel contained in the kit 11 kinds of reagents with 02 controls, 02 ml positive one (Test EP histamine 10 mg/ml) and another negative (Test EP saline) to 03 ml and the other nine types Dermatophagoides pteronyssinus 20%, Dermatophagoides farinae 20% Blomia tropicalis 5%, Alternaria alternata 5%, Aspergillus fumigatus 5%, Cockroach 5%, Mosquito 5%, Dog epithelium 5%, Cat epithelium 5%, each bottle with 3 ml. The kit was kept as standard, from 2 to 8 ℃, and its validity was until 01.31.2016.
For the test the pre-cleaning of the skin was performed in the ventral face average (volar) forearms, with running water and soap degermante commonly used in healthcare facilities. ID (13 × 3.8), single use, later discarded in an appropriate place an intradermal sterile needle was used. After holding the first pick in the skin with the needle was marked the limit of at least 3 cm between the locations of the other picks. The needle was inserted perpendicular to the skin at a small pressure. Subsequently, a drop of controls for comparison with other reagents was placed. After each pick, respecting the space defined earlier, the above antigens were put through a dropper forbade each vial individually, with no contamination by other reagents. It determined also the distance of at least 1 cm for the application, with no contamination of the dropper by the skin. 15 minutes after application of the last reagent, the reading of the papules formed was carried out which should be at least 3 mm in diameter to be confirmed as a positive reaction to the reagent, and recorded in a proper form. At the end of the readings was asked participants to wash the place with water and dried up with paper towels. Patients were alerted to the persistence of itching, swelling and redness at the pick of the place, being discouraged from using any kind of ointment, cream, lotion or product that is not prescribed by the doctor as well as to avoid scratching or rubbing [17-20].
Children and adolescents using systemic allergy Topics presence of lesions and dermatitis at the site of the test; anaphylaxis history; crisis of asthma and rhinitis in before the test day or 5 days and the presence of fever were free of tests.
For the test FeNO was used NIOX MINO® (Aerocrine AB, Solna, Sweden) as recommended by the ATS [21] and by the manufacturer. The exhalation flow rate was 50 ml/s, after expiry of the test time until the reading from 5 to 30 seconds reading time of about 34 s [22]. 3 measurements on each participant, with 10-minute interval between them were conducted. In the final analysis, it was considered the average of the measurements. In interpreting the values was performed using the parameters of Table 1 as indicated by ATS [21] and the manufacturer [23]. The patients rest 15 minutes before the test. In previous contact patients were instructed to make them fast for a minimum of one hour. The tests were performed in a standing position.
Table 1: Hay reference values according to age. View Table 1
The asthma control test (ACT) was applied to all queries, always by the same interviewer. The ACT has five items that relate to the symptoms, rescue medication use and effect of asthma on daily activities without the need to function measurements pulmonar. Each question has an answer scale whose score ranges from 1 to 5, resulting in a total score test between 5 and 25 points. The score of 25 refers to total control. The score between 20 and 24 points indicates partially controlled asthma, and a score below 20 means uncontrolled asthma, below 20 means uncontrolled asthma [24-26].
For spirometry was used machine (VM1 ATS, Clement Clarke, CE0120, Made in the U.K.), with disposable polypropylene nozzle in the standing position and always by the same observer. The technical procedures, the criteria for acceptability and reproducibility followed the standards of the ATS/ERS. The parameters analyzed were forced vital capacity (FVC), forced expiratory volume in one second FVC (FEV1) and Tiffeneau index (FEV1/FVC). Children and adolescents were instructed to perform a maximal inspiration and then exhale all the air volume with maximum effort with his mouth properly attached to the mouth, avoiding air leakage. For proof bronchodilator salbutamol have been used four jets (Aerolim® - 100 g/jet), with a minimum interval of one minute between them. After 15 minutes the test was repeated and the bronchodilator response was considered positive for values over 12% in FEV1 (% predicted) [1]. For the predicted values were used the equations of Polgar and Promadhat [27].
The study included patients of both genders, with persistent asthma or persistent moderate and severe, classified according to the criteria of the GINA (2015), with no history of treatment with inhaled corticosteroids 30 days before the first visit (baseline), at the age of 7-15 years old, and able to perform spirometry and measurement of FeNO. Exclusion criteria were chronic diseases besides asthma, recent airway infections, presence or history of fever in the last 30 days, exacerbation of asthma, smoking and inability to perform the proposed procedures.
All procedures were conducted in accordance with the Declaration of Helsinki and with the approval of the Institutional Ethics Committee (0004.0.418.000-11).
Variables were described as measures of central tendency (mean) and variability (standard deviation). To normalize the data, we used the Kolmogorov-Smirnov normality test. Comparisons of the variables of pulmonary function testing, pre- and post-bronchodilator, were performed using the paired t test. For longitudinal comparisons of variables was used One Way ANOVA or Kruskal-Wallis when indicated. To analyze the associations between different variables and between the same variable, according to the visits, we used the Pearson or Spearman test when indicated. The FeNO was categorized and analyzed by age group and their comparison between visits were not normally distributed and the Kruskal Wallis test and Mann Whitney tests were used. Data analysis was performed with the Graphpad Prism software version 6.0.O level of significance was considered at p < 0.05.
Thirty-seven children and adolescents eligible for the study, between January and November 2015, 22 participated in three visits and completed the proposed procedures. Fourteen of them appeared in only one query and/or failed to perform the proposed tests (FeNO and spirometry). A child dropped from the study.
Table 2 show the characteristics of the 22 patients in the study. There was no difference in the number of participants divided by gender. In regard to race it has been observed that most participants consisted of brown (54.5%), white (36.4%) and black (9.1%). In allergy testing was predominantly atopic asthma (72.7%). In the classification of asthma severity was observed that 72.7% of participants had persistent and moderate asthma, and only 27.3% severe. The bronchodilator test was negative in 77.3% of patients with asthma.
Table 2: Sample characterization. View Table 2
Data from the longitudinal evaluation of spirometry, FeNO, the control tests of asthma and the symptoms are shown in Table 3. For the presentation of FeNO results, patients were divided into subgroups for those aged < 12 years and ≥ 12 years and later into low, intermediate and high values of this variable as the standardization of the manufacturer. The mean score of the ACT was 19:36 ± 3.27 at baseline, 20.0 ± 4:49 and 20.77 ± 4.02 in the second and third visits, respectively. However, the increase was significant only between the second and third visit (p = 0.0309). The TCA correlated moderately and positively with FEV1 % at baseline (r = 0.4480, p = 0.0417). In relation to data in lung function, FEV1 (%) did not vary significantly between visits. Otherwise, the FEV1/FVC ratio (%) significantly increased basal query to the third visit (p = 0.0098). Levels of FeNO, FEV1 (%), the FVC, FEV1/FVC ratio (%), and ACT scores did not differ between atopic and non-atopic subgroups in 3 time points.
Table 3: Longitudinal evaluation data of the fraction of exhaled nitric oxide of the control tests of asthma and lung function and symptoms reported in the consultations. View Table 3
Reported symptoms shown in Table 4 indicate a predominance of cough complaint, both daytime as night with little variation between appointments, but with a slight decrease compared with the baseline visit 3 (10/13 to 8/8). Chest pain was unreported in both the basal (04 participants) as the third visit (03 participants). Table 4 show the mean ± SD of the absolute values according to the age and levels of severity of FeNO.
Table 4: FeNO levels according to age and their respective benchmarks. View Table 4
The FeNO correlated significantly between the banal and the second visit, as well as the second and third visits (Figure 1). As expected FeNO correlated inversely with Tiffeneau index assessed at baseline as well in the second and third visits (Figure 2).
 Figure 1: Relationship of fraction of exhaled nitric oxide (FeNO) in basal visits, 2nd and 3rd visits.
View Figure 1
Figure 1: Relationship of fraction of exhaled nitric oxide (FeNO) in basal visits, 2nd and 3rd visits.
View Figure 1
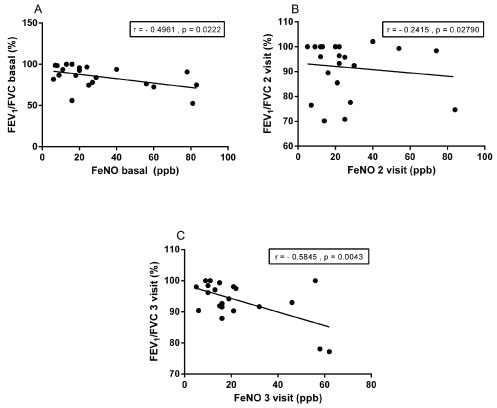 Figure 2: Relationship of fractional of exhaled nitric oxide (FeNO) and Tiffeneau index, the baseline visits, 2nd and 3rd visits.
View Figure 2
Figure 2: Relationship of fractional of exhaled nitric oxide (FeNO) and Tiffeneau index, the baseline visits, 2nd and 3rd visits.
View Figure 2
The ACT score of Baseline was positively correlated with FEV1 % predicted from the baseline visit (r = 0.4480, p = 0.0417) in Figure 3.
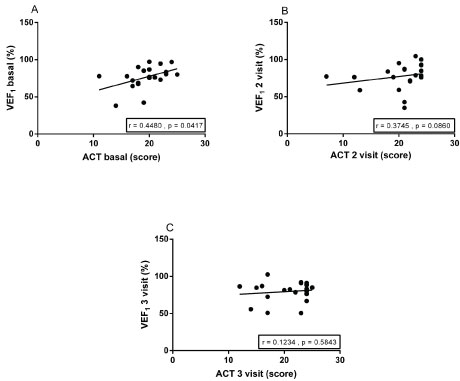 Figure 3: Correlation of the asthma control test (ACT) and forced expiratory volume in the first second of forced vital capacity (FEV1), the baseline visits, 2nd and 3rd visits.
View Figure 3
Figure 3: Correlation of the asthma control test (ACT) and forced expiratory volume in the first second of forced vital capacity (FEV1), the baseline visits, 2nd and 3rd visits.
View Figure 3
Drug therapy of asthma is the cornerstone of treatment and the optimal use of medicines can, in most cases, help patients control the symptoms and reduce the risk of future morbidity. Therefore, the evaluation and monitoring should be performed regularly to reassess and adjust the treatment [1]. Due to the heterogeneity of asthma, multiple biomarkers are required for the diagnosis and management [9]. This study evaluated the effects of beclomethasone on lung function, airway inflammation and asthma control in children and adolescents in three consecutive visits with an interval of two months between them. Lung function, as measured by the ratio FEV1/FVC (%) increased significantly from the first to the third visit. Furthermore, it was observed that the level of exhaled inversely correlated with the increase in the ratio FEV1/FVC (%) evaluated in three visits. The level of asthma control, as assessed by ACT, positively correlated with FEV1 %, only on the first visit. The FeNO levels, FEV1 (%), FEV1/FVC (%) and the ACT score did not differ between participants with atopic asthma, non-atopic, defined by a positive skin test for all three times.
In the classification of asthma severity was observed that 72.7% of participants had mild persistent and persistent asthma, and only 27.3% severe persistent. The level of asthma control participants consisted primarily of partly controlled asthma (45.5%) and non-controlled (50%). In the third visit, the proportion of participants with partially controlled asthma increased to 63.63% and uncontrolled decreased to 31.31%, suggesting an increase in asthma control from the beginning of the introduction of corticosteroids for asthma control. The predominant symptom in this sample was coughing complaint, both daytime as night with little variation between appointments, but with a slight decrease comparing the first and third visit. Chest pain was rarely reported during treatment.
Inhaled corticosteroids have been considered as first-line treatment in adults and children who have persistent asthma. The corticosteroids suppress inflammation by positively affects the airway inflammation and asthma symptoms in most patients. Its main action is to inhibit the inflammatory genes encoding cytokines, chemokines, adhesion molecule, enzymes and inflammatory receptors [28].
One of the strategies used to monitor asthma control and response to anti-inflammatory agents such as corticosteroids, is the assessment of the level of exhaled nitric oxide, a marker of airway inflammation. The FeNO was the first and is the most widely marker inflation airway used [9]. According to some authors the level of FeNO predicts the likelihood of responding to corticosteroids more consistently than spirometry, bronchodilator response, variability of peak expiratory flow (PEF) and hiperresponsividade to methacholine [29-31]. Other authors have shown that FeNO can be used as a marker of asthma control [21] and that this present rapid response to treatment modification [32]. However, studies using FeNO as an alternative method to modify treatment with corticosteroids based on symptoms and/or pulmonary function are conflicting [33]. For the analysis of the exhaled results, patients in this study were divided into two subgroups, age ≥ 12 and ≤ 11 years, according to the cutoff point recommended by the manufacturer. Levels of FeNO were significantly higher in children aged ≥ 12 years (48.9 ± 31.1 ppb) compared to those aged ≤ 11 years (21.7 ± 16.1 ppb). However, both values were classified as intermediaries in their respective populations. It was also observed that the dispersion of FeNO values was high in both age groups, in all visits. These results are in agreement with data [34] evaluating asthmatic children and adolescents (7 - 17 years) and found that FeNO levels ranged from 17 to 56.2 ppb. No present study was also observed that FeNO correlated significantly between the three measures.
Several authors have shown that the exhaled nitric oxide levels are high primarily in atopic asthma [9,35,36]. In this study, FeNO levels did not differ between participants with atopic asthma, non-atopic, the three conditions. In contrast, [36] demonstrated that atopic asthmatics with moderate persistent asthma and severe (6-17 years) and use of anti-inflammatory medication for at least one year, have higher levels of exhaled nitric oxide, suggesting the persistence of inflammatory activity despite the use of anti-inflammatory medication. Similar to the data presented by [34], a wide dispersion was observed the FeNO levels at every visit, regardless of the age range studied. The wide dispersion of FeNO levels and the small number of participants may explain the difference in results. As can be seen in Table 2, the number of asthma patients with low levels of exhaled increased from 9 to 16 patients, considering the first and third visits. Moreover, the number of asthma patients with intermediate levels of exhaled reduced from 8 to 2 patients, given the same period. As the number of patients with high levels of exhaled remained constant throughout the visits, these results suggest that treatment with corticosteroids decreased airway inflammation in this population.
In relation to data in lung function was observed that FEV1 (%) did not vary significantly between visits. Otherwise, the FEV1/FVC ratio (%) significantly increased basal query to the third visit, suggesting improvement in lung function. Unlike many authors [34,37] that found no association between reduction in FeNO levels with improved lung function in this study the FeNO levels inversely correlated with the Tiffeneau index, assessed at baseline as well in the second and third visits. These results suggest reduction of inflammation was accompanied by an improved pulmonary function. Our results corroborate those of Yao, et al. [38] who observed correlation between levels of FeNO and FVC, FEV1 and FEV1/FVC. FEV1 levels (%), the FVC, FEV1/FVC ratio (%), and ACT scores did not differ between atopic and non-atopic subgroups in 3 time points.
This study has some limitations. Due to the small sample size, the FeNO levels were not measured in sub-groups according to gender. Hervás, et al. [34] and Zhang, et al. [39] demonstrated that FeNO levels in males are higher than females. Another limitation was the selection of patients. In this study the prevalence of diseases related to atopy has not been established. Levels of FeNO may appear to change in various conditions such as rhinitis and atopic eczema, leading to difficulty in interpreting the results. Finally, the cutoff used in this study to establish the limits of normality, were those recommended by the manufacturer. However, this inflammatory marker, and is modulated by several factors such as age, gender, height, ethnicity, heterogeneity of asthma phenotypes and diseases related to atopy, it is also modulated by environmental factors (smoking, environmental pollution, the measurement time, season) [9,40].
This study showed that treatment with corticosteroids improved the classification of the level of asthma control and that despite the great dispersion of the FeNO levels, it correlated inversely with Tiffeneau index at the three assessments.
This work was supported by the Fundação Lucas Machado (FELUMA), Faculdade Ciências Médicas-Minas Gerais, Pós Graduação em Ciências da Saúde and Pneumology Pediatric Clinic of Unidade de Referência de Saúde Campos Sales, da Prefeitura de Belo Horizonte (URS Campos Sales- SUS BH). We are thankful to Isabel Cristina Gomes for statistical analysis assistance.
No conflict of interest exist.
UOA- Conception and design, acquisition of data, analysis and interpretation of data, drafting the article, revised and approved final version of manuscript; GSM- Analysis and interpretation of data, edited and revised manuscript, approved final version of manuscript; NSJ- Conception and design, analysis and interpretation of data, drafting the article, revised and approved final version of manuscript; MGRM- Conception and design, analysis and interpretation of data, drafting the article, revised and approved final version of manuscript.