Spontaneous coronary artery dissection (SCAD) is a rare cause of acute myocardial infarction (MI) and sudden cardiac death (SCD). SCAD is mostly described in young females with a sparse cardiac history. The pathophysiology of SCAD has not been clearly elucidated in the literature and the underlying etiology continues to be investigated.
We describe a unique case of a 57-year-old female who presented with chest pain who was found to have a ST segment elevation myocardial infarction (STEMI) on electrocardiogram. Cardiac coronary angiography demonstrated SCAD of the mid to distal left anterior descending (LAD) artery who was originally treated with, but ultimately failed, medical management. Less than 24 hours after presentation, she had a ventricular fibrillation cardiac arrest with repeat coronary angiography indicating complete occlusion of her mid to distal LAD, resulting in management with coronary artery bypass grafting (CABG).
Conservative management of SCAD should be attempted with patients however LAD lesions are at high risk for cardiac decompensation, requiring invasive revascularization with PCI or CABG.
SCAD: Spontaneous Coronary Artery Dissection; MI: Myocardial Infarction; SCD: Sudden Cardiac Death; STEMI: ST Segment Elevation Myocardial Infarction; LAD: Left Anterior Descending; CABG: Coronary Artery Bypass Grafting; PCI: Percutaneous Coronary Intervention; ECG: Electrocardiogram; ROSC: Return of Spontaneous Circulation; ACS: Acute Coronary Syndrome; CA: Coronary Angiography; OCT: Optical Coherence Tomography; CCTA: Coronary Computed Tomography Angiography; ACEi: Angiotensin-Converting Enzyme Inhibitors; ARB: Angiotensin-Receptor Blockers
Spontaneous coronary artery dissection (SCAD) is defined as an intramural hemorrhage leading to the separation of the layers of the coronary artery wall with or without an intimal tear [1]. SCAD is a rare, but life threatening event typically seen in young females between the ages of 47-53 [1-5]. The disease is often associated with peripartum myocardial infarctions, however only about 15-20% of peripartum myocardial infarctions are due to SCAD [6], while approximately 10% of overall SCAD cases are associated with pregnancy [7,8]. Acute myocardial infarctions due to SCAD account for less than 1% of cases [1,3,9]. Patients who develop SCAD have a lower incidence of typical cardiac risk factors when compared to those with myocardial infarctions from atherosclerotic disease [1,3,9-11]. SCAD has been historically reported to be associated with an inciting event, often an emotional stressor or hormonal triggers. There is a high prevalence of fibromuscular dysplasia with purported incidence as high as 50% of patients with SCAD [12-14].
Due to the low incidence of SCAD, there is a paucity of data regarding an effective management algorithm. As this is not an atherosclerotic disease process, SCAD is managed differently than typical myocardial infarctions. We describe in this case report how SCAD management is highly dependent on hemodynamic stability, anatomical features, and severity of patient symptoms ultimately including both medical management and revascularization with CABG.
The patient is a 57-year-old female with a past medical history of paroxysmal supraventricular tachycardia, hypertension, and hyperlipidemia who presented to the emergency department with one hour of substernal chest discomfort that radiated to both arms. Electrocardiogram (ECG) demonstrated ST elevation in the anterior and inferior leads with lateral reciprocal changes. She was taken for an emergent coronary angiography, which demonstrated a dissection of the mid to distal LAD artery (Figure 1). In the cath lab, the patient had TIMI II flow and was treated with intracoronary nitroglycerin and diltiazem, which complete resolution of symptoms (Video 1). She was managed post-procedure in the intensive care unit with a heparin infusion, aspirin, clopidogrel, nitroglycerin, metoprolol, atorvastatin, olmesartan, and hydrochlorothiazide. Echocardiogram at that time demonstrated normal to mild decrease in left ventricular function with a left ventricular end diastolic pressure of 24 mmHg. With conservative management the patient was hemodynamically stable and asymptomatic after angiography. The following day, the patient had a sudden cardiac arrest with sustained ventricular fibrillation. Return of spontaneous circulation was achieved after 1 milligram of IV epinephrine and one defibrillation with 150 J. Given the brevity of ACLS, she did not require intubation as she regained consciousness immediately after the event. ECG post-cardiac arrest demonstrated ST elevations in leads V2 to V4. The patient was emergently sent to the catheterization lab for repeat coronary angiography this time demonstrating an occluded LAD that was not amenable for revascularization due to distal occlusive thrombus and extensive dissection (Video 2). Patient was treated with an emergent CABG of the left internal mammary artery to the LAD. Following surgery, the patient required two units of packed red blood cells, two units of platelets, and four units of fresh frozen plasma. The patient was stabilized and asymptomatic postprocedure and was discharged on postoperative day 5 with return to baseline function.
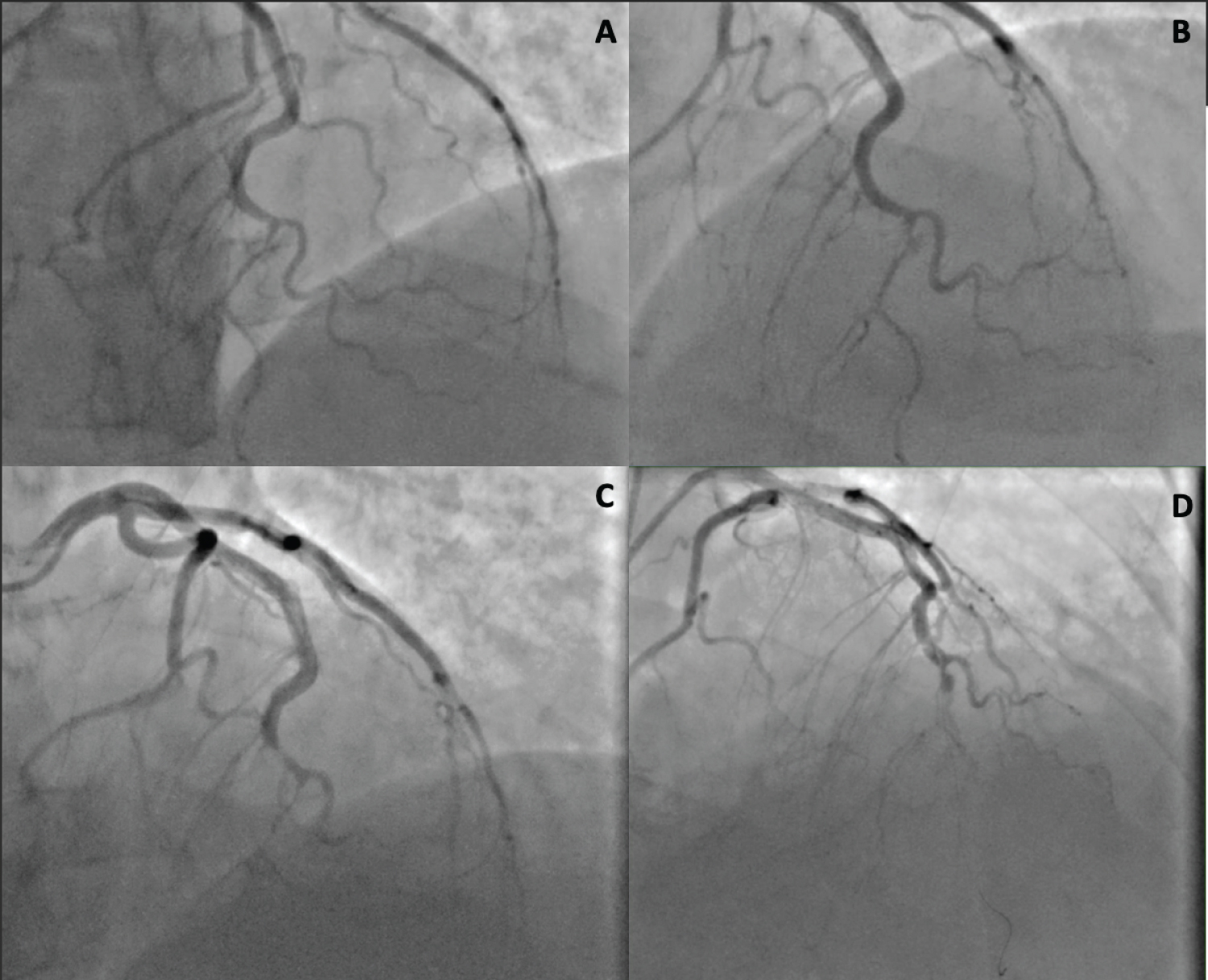 Figure 1: A and B is the initial cardiac catheterization demonstrating a SCAD; C is the emergent cardiac catheterization after cardiac arrest demonstrating distal occlusive thrombus of the distal LAD due to SCAD; D illustrates an attempt at percutaneous coronary intervention with stenting which was unsuccessful.
View Figure 1
Figure 1: A and B is the initial cardiac catheterization demonstrating a SCAD; C is the emergent cardiac catheterization after cardiac arrest demonstrating distal occlusive thrombus of the distal LAD due to SCAD; D illustrates an attempt at percutaneous coronary intervention with stenting which was unsuccessful.
View Figure 1
SCAD accounts for < 1% of all acute myocardial infarctions [1]. Shah, et al. found that the prevalence of SCAD is between 0.1-1.1% [15,16]. In addition, Antonutti, et al. found that SCAD accounts for 0.5% of sudden cardiac deaths [17].
Studies have demonstrated that SCAD is not typically associated with the prototypical risk factors of ACS including hyperlipidemia, hypertension, diabetes [16]. Kim found that SCAD is associated with several risk factors including pregnancy and hormonal changes, emotional stress, physical stress, use of stimulant medications, illicit drugs, and inflammatory disorders such as systemic lupus erythematosus, Sarcoid, inflammatory bowel disease, and celiac disease [1]. Shah, et al. were able to determine that atherosclerosis, vigorous exercise and coronary vasospasms are also risk factors for SCAD [1,16].
SCAD can be divided into two pathologic subsets: 1. SCAD with an intimal tear and 2. SCAD with no intimal tear [18,19]. Studies have hypothesized that an intimal tear leads to bleeding into the vessel wall which in turn allows communication between a true lumen and false lumen resulting in compression of the true lumen by elevated pressures in the false lumen [19]. Conversely, SCAD without an intimal tear is hypothesized to develop a disruption of the vasa vasorum resulting in the formation of a hematoma producing an occlusion of the coronary lumen leading to an ischemic event [19,20].
Clinical features: Lettieri, et al. demonstrated that SCAD manifests as a myocardial infarction in 90% of patients. Several studies have demonstrated that a ST elevated myocardial infarction is associated with 20 to 50% of cases [1,2,21,22], while 3-5% of cases present with ventricular arrhythmia [1,2,21] and approximately 2% of cases will present in cardiogenic shock [21]. The chief complaint of patients presenting with SCAD is typically chest pain that radiates into the arm, neck or back, dyspnea and diaphoresis [1,23,24]. Tweet, et al. found that the majority of patients with SCAD present with ACS symptoms including chest pain radiating into the left arm, neck, back, epigastric pain, nausea, vomiting, diaphoresis or dyspnea [25,26]. While SCAD manifests much like an MI, a high index of suspicion should be warranted despite normal troponin levels. Lindor, et al. elucidated that approximately 27% of initial troponin levels in patients with SCAD will be reported as normal [24]. However, other studies have shown that cardiac enzymes are elevated in > 95% of cases [2,11,25,26]. In addition to physical symptoms, Boulmpou, et al. found that precipitating stress factors including emotional tension or previous excessive physical activity are reported [25]. Saw, et al. showed that SCAD can be complicated by ventricular arrhythmias, cardiogenic shock or sudden death [2]. In summary, young women with symptoms of ACS with few or no risk factors should have a high index of suspicion for SCAD. According to a review done by Afzal, et al. early detection is imperative to manage SCAD and decrease the mortality and morbidity rates [16,27].
Diagnostic tests/Imaging
Coronary angiography: Gold standard diagnostic technique for SCAD is performing an emergent coronary angiography (CA) [1,10,16,18,28,29]. CA allows for the ability to assess the extent of the disease and potential for angioplasty [1]. Guragai found that CA allows for visualization of the characteristic signs of the conditions and that diagnosis is often supplemented by intracoronary/intravascular ultrasound [28].
The two patterns of SCAD--intimal tear vs. no intimal tear--can be identified by coronary angiography. SCAD initiated by an intimal tear can be visualized as multiple lumens, an intimal flap, or slow clearing of the contrast dye [18,29]. SCAD with no intimal tear will present mimicking atherosclerosis, with hematoma formation manifesting as narrowing or occlusion of the coronary artery [10,18,29]. While the gold standard diagnostic technique for SCAD is CA, oftentimes it can be missed and further diagnostic techniques can be utilized if the index of suspicion is high. Intravascular imaging, particularly optical coherence tomography, and coronary computed tomography angiography are utilized in cases where CA is non-diagnostic despite clinical suspicion [1,15].
Optical coherence tomography (OCT): OCT is an advanced intravascular imaging system that allows for accurate visualization of the coronary artery wall with a high axial spatial resolution of 10-20 micrometers [1,10,19,30,31]. OCT is a rotating fiber-optic system with which infrared light can be detected and reflected within the tissue to create a high resolution detailed image of the coronary vessels. Kim, et al. found that use of OCT may avoid unnecessary and harmful intervention for SCAD, while providing an opportunity to tailor treatment for patients with SCAD [1]. Alfonso, et al. performed a prospective study assessing the value of OCT and found that it was successful in ruling out diagnosis of SCAD with images that mimic SCAD and where angiography has limited value [19]. OCT is useful in diagnosis of SCAD by allowing the visualization of the double-lumen morphology, identifying the entry tear, characterizing the circumferential and longitudinal extent of disease and branch involvement, and identifying the presence of an associated thrombus [10,19]. In addition, Kim elucidated OCT can identify true and false lumens, intramural hematomas, dissection flaps, fenestrations and entry tears connecting true and false lumens [1].
Although OCT has several advantages in diagnostics of SCAD, it comes with a risk. According to Jackson, et al. 8% of patients undergoing OCT may develop associated complications including dissection extension, impaired flow after acquisition of imaging, iatrogenic dissection, and cannulation of false lumen [32]. Therefore, the gold standard of diagnosis remains CA. OCT is becoming a vital diagnostic modality when CA assessment is nonspecific or nondiagnostic in the setting of elevated clinical suspicion.
Intravascular ultrasound (IVUS): IVUS is an imaging modality that is utilized in SCAD. It has an axial resolution of 150 um which allows for the identification of intraluminal thrombi, intramural hematoma, intimal tear and atherosclerotic plaque [33]. In comparison to OCT, IVUS has an increased depth of penetration and allows for visualization of all three layers of the vessel wall allowing for delineation of the true lumen from false lumen in SCAD [34]. Additionally, the increased depth of penetration allows for improved visualization of the dissected segment even in the setting of larger arteries or in the presence of a thrombus [34]. While IVUS certainly is advantageous in diagnosis of SCAD, its limitation is poor spatial resolution which deems it unreliable in identifying the site of communication between true and false lumen [35]. According to Paulo, et al. this flaw is mitigated through the utilization of combined OCT and IVUS imaging for the evaluation and management of SCAD.
Coronary computed tomography angiography (CCTA): CCTA is a noninvasive diagnostic modality that can be utilized to diagnose SCAD most commonly in the setting of uncertain CA [1,10,15]. CCTA can be used to visualize dissection flaps, stenoses, and intramural hematomas [1,36]. In addition to being able to visualize the details of the SCAD, CCTA allows for an evaluation of the coronary vessels without the risk of complications including dissections, arrhythmias, myocardial infarction and femoral artery injury that can occur with CA and OCT [37]. While CCTA has the advantage of being non-invasive, it has limitations. Atherosclerotic plaques that have not calcified may appear as intramural hematomas and the limited resolution for small vessels prevents accurate visualization of the distal portion of the coronary vessels, which are most commonly affected by SCAD leading to a high rate of false negative studies [38,39]. A common population affected by SCAD includes pregnant females, for which CCTA would result in induction of radiation for both mother and highly developmental fetus making CCTA relatively contraindicated [10].
Management of SCAD is different from the management of atherosclerosis due to 3 major factors which are the underlying pathological features, the challenge of PCI, and the majority of medically treated SCAD demonstrated angiographic evidence of healing over time [1]. Due to the limited incidence of SCAD, there is a paucity of randomized control trial (RCT) data surrounding the appropriate treatment model for SCAD. Treating a patient with an acute MI due to SCAD with medical therapy or revascularization is dependent on several factors making it a complex decision [1]. Clinical status, vascular territory at risk, and degree of stenosis of the affected vessel are aspects to consider when deciding between the two. Multivessel proximal dissections, left main or left anterior descending artery involvement are considered high risk features warranting a discussion of surgical intervention [15,33,40]. In addition patients with hemodynamic instability, shock or life threatening arrhythmias are considered for immediate intervention. Thrombolysis in Myocardial Infarction (TIMI) is another factor that is considered in definitive management of a patient with SCAD. Tweet, et al. advocate for avoiding revascularization in patients with SCAD who are in stable condition with sufficient distal vessel perfusion despite clinically significant stenosis [22]. Hayes, et al. suggest immediate revascularization is necessary for patients with high risk features with PCI or CABG. Majority of SCAD cases can be treated conservatively with medical management. Post-medical management angiography of patients with SCAD demonstrates healing over time with restoration of blood flow and a decrease in stenosis [1]. Thus 80% of patients can be successfully treated medically and this is preferred by expert consensus, due to limited RCT data, over revascularization [2,15,33].
A majority of case reports demonstrate that medical management is preferred in patients in stable condition which is how our patient was initially managed. Our patient had the clinical signs and symptoms of a myocardial infarction and was appropriately taken to the cath lab at which point the diagnosis of SCAD was determined. She was appropriately managed medically with complete resolution of symptoms and stable hemodynamics. Unfortunately, the patient decompensated rapidly, became hemodynamically unstable and had a cardiac arrest followed by successful resuscitation. At this point, due to her hemodynamic instability and high risk features including LAD lesion and cardiac arrest, she was taken to the cath lab for emergent revascularization. D’Ovidio, et al. reported that PCI can be used to manage SCAD with excellent results, but risk of stent placement in the false lumen has been noted frequently [10]. While our patient was attempted for a PCI, stenting was unable to be achieved and the patient was taken for an emergent CABG. Mortensen, et al. illustrated that CABG is indicated in patients with main stem, multivessel, left main coronary artery involvement, failed medical or PCI therapy, hemodynamic compromise or luminal narrowing [11]. Our patient not only had left main coronary artery involvement with luminal narrowing, but also failed medical and PCI therapy in addition to developing hemodynamic compromise indicating the utilization of CABG. This successfully treated our patient who was later discharged home post-operatively.
Medical management: Conservative management of SCAD is done similarly to the treatment of an MI due to atherosclerosis. Patients are initiated on anticoagulation and dual antiplatelet therapy, however once SCAD is confirmed, anticoagulation is discontinued due to the risk of worsening the intramural bleed [1]. Thrombolytic medications are also contraindicated in the management of SCAD due to bleeding risk [41]. Dual antiplatelet therapy is considered beneficial in patients with SCAD because of the prothrombotic state of the intimal tear and formation of intramural hematomas [25]. In addition to dual antiplatelet therapy, patients are started on beta-blockers, statins and angiotensin-converting enzyme inhibitors (ACEi) or angiotensin-receptor blockers (ARBs).
Beta-blockers have been shown to have a preventive effect in the recurrence of SCAD. Saw, et al. found that the use of beta-blockers resulted in a 64% decrease in the incidence of recurrent SCAD over the course of 3 years [42]. Suzuki, et al. elucidated that beta-blockers seemed to reduce arterial stress, preventing further dissection, a principle originating in the treatment of aortic dissections [43]. Martins, et al. illustrated that beta blockers have the additional potential to facilitate healing and reduce shear stress placed on arteries thus preventing true lumen patency and thrombotic occlusion [44]. Similarly, ACEi and ARBs are used in patients with SCAD if left ventricular dysfunction is present or when the ejection fraction falls under 40% and statins are prescribed in cases of hyperlipidemia [1,15].
Operative: Operative management of SCAD includes PCI and CABG. PCI in patients with SCAD is challenging and success rates range from 47% to 72% [2,22,40]. Complications from using PCI include the use of multiple stents in the case of long lesions, coronary wires entering the false lumen causing vessel occlusion, iatrogenic injury and loss of -distal-vessel patency or retrograde extension to more proximal vessels due to hematoma progression [45]. An adverse effect seen in long-term follow up of SCAD patients being treated with PCI include stent malposition due to resorption of the intramural hematoma over time [19,46]. One burgeoning alternative with limited case studies is the use of cutting balloon angiography (CBA), which causes fenestration in the false lumen to allow for communication back from the false lumen to the true lumen distal to the lesion [47]. Due to limited randomized control trial data, long-term outcomes, and limited case reports, the utilization of CBA is not widely accepted. There are several factors associated with deciding to use CBA for SCAD. The dissected wall of the artery is frail, and thus susceptible to perforation limiting CBA to small diameter balloons and low inflation pressures. According to Main, et al. CBA should be avoided in vessels < 2 mm in diameter and vessels which are not tortuous [47]. In addition, utilization of CBA in the left main has not yet been studied and is associated with increased risk due to the size of the vessel. Current expert consensus for SCAD recommend lesions involving the left main require CABG for optimal therapy [41]. While outcomes for PCI and CABG are still debated for SCAD, this novel therapy of CBA needs to be studied through randomized control trials to evaluate its efficacy in managing SCAD with failed medical therapy.
While PCI may be done successfully, complications make it a challenging procedure. Meanwhile, CABG for revascularization has been successful [22]. CABG is reserved for SCAD involving high-risk lesions involving the left main or left anterior descending artery where attempts at PCI have failed, or in those whose large areas of myocardium are at risk and medical therapy alone is unable to treat the ischemia [1,22]. According to Shah, et al. CABG is the preferred treatment for hemodynamically unstable patients and first line in patients with large left main stem dissections [16]. Almaddah, et al. showed that in addition to hemodynamically unstable patients and left-main dissections, patients with ventricular arrhythmias are also candidates for CABG [48].
While there is a paucity of data regarding the treatment of SCAD, our case report uniquely demonstrates both conservative and surgical management outcomes for a patient with SCAD. Our recommendation is to attempt conservative management with close monitoring for possible conversion into a high risk case. In addition, SCAD of the LAD should be even closely monitored due to the vascular territory at risk in the case conservative management fails. High risk SCAD should be evaluated for emergent revascularization with PCI or CABG. Close monitoring, early recognition and prompt surgical management with PCI/CABG lead to a successful outcome.
Consent was obtained to publish this case report.
None.