Pelvic inflammatory disease is extremely rare during pregnancy. Both differential diagnosis and management of tubo-ovarian abscesses in pregnancy are challenging and associated with poor obstetric outcomes. The authors present a case of a 17-years-old girl with low abdominal pain for five days. A tubo-ovarian abscess was suspected, with an early pregnancy of unknown location. She was admitted to receive intravenous antibiotics and to rule out ectopic pregnancy. There was clinical and analytic recovery and intrauterine pregnancy was confirmed. At the tenth week of pregnancy she was readmitted with spontaneous abortion. This case highlights how challenging first-trimester low abdominal pain may be, as well as the probable effect of medications during early pregnancy.
Tubo-ovarian abscess, Pelvic inflammatory disease, Female pelvic pain, Early pregnancy, Pregnancy of unknown location
A case of pelvic inflammatory disease with tubo-ovarian abscess in early pregnancy and its management are reported.
Pelvic Inflammatory Disease (PID) is an inflammatory and infectious disorder of the female upper genital tract. PID is caused ascendant infections from the inferior part of the genital tract through the cervix to the uterus and the Fallopian tubes. Most of the times it is caused by multiple bacteria, in most of the cases sexually transmitted infections including Chlamydia trachomatis, Neisseria gonorrhoeae, Gardnerella vaginalis, Haemophilus influenzae, and anaerobes. The diagnosis is based on clinical findings, serum analyses and pelvic ultrasound or CT scan. The main signs and symptoms are low abdominal pain, purulent cervical discharge, cervical tenderness, dyspareunia and hyperthermia but patients may be completely asymptomatic. There is usually elevation of the inflammatory serum parameters, such as leucocytosis with neutrophilia and elevated C-reactive protein (CRP). At ultrasound there may be no abnormal finding or they may be images suggestive of tubo-ovarian abscesses (TOA).
One of the main differential diagnoses is ectopic pregnancy as this may also present as low abdominal pain, adnexal mass and free pelvic fluid. Pregnancy and PID are rare to occur simultaneously in such a way that there is no estimated prevalence of this disease during pregnancy in the literature. It is still not clear how pregnancy, a physiological status of immunosuppression, may confer some kind of protection against ascending pelvic infections. Pregnancy itself is thought to provide a barrier preventing the ascent of bacteria into the uterine cavity [1]. Cervical mucus may also have a role. The treatment of PID is antibiotic therapy [2,3].
There are many causes of female low abdominal pain, pregnancy (intrauterine or ectopic) and pelvic inflammatory diseases are frequent ones, but usually only one of these diagnoses is considered. If there is an inflammatory status with hyperthermia, vaginal discharge, cervical tenderness and large heterogeneous adnexal masses, it is suggestive of PID. On the other hand, if one has a positive pregnancy test or high beta hCG serum levels, the diagnosis of pregnancy is assumed and PID is quite improbable [4,5].
Another challenging point is the management of established PID in a pregnant patient. PID is thought to be associated with poor obstetric outcomes, including impaired embryo implantation, miscarriage, infection, foetal demise, premature rupture of membranes (PROM) and preterm labour. Furthermore, the safety of most of the antibiotics in pregnancy is not clear, especially during the first trimester. There is also little information about surgical procedures (especially those concerning the pelvic organs) during early pregnancy [4,5].
A 17-year-old primigravida female with no significant medical history presented to the emergency room with pelvic pain for the past five days. She reported having started her sexual activity one year before, with two sexual partners until date, the last relationship was recent, and she denied the use of any contraceptive method. She had amenorrhea of three weeks and four days. There were no other symptoms. At general examination she was apyretic, with normal vital signs. She presented pain at low abdominal palpation but there were no signs of acute abdomen. She also presented a purulent and odorous cervical discharge and cervical tenderness.
Serum analysis revealed slight anaemia (11.2 g/dL), leucocytosis (18.2 × 103/uL) with neutrophilia (13 × 103/uL, 71%) and elevated CRP (117.3 mg/L). Beta hCG was 113.6 mUI/mL.
Transvaginal ultrasound scan revealed a thickened double layer endometrium with 17 mm but empty uterine cavity, together with a heterogeneous adnexal mass at the right tube with 6 cm of largest diameter and a small amount of free fluid at the Douglas Pouch (Figure 1).
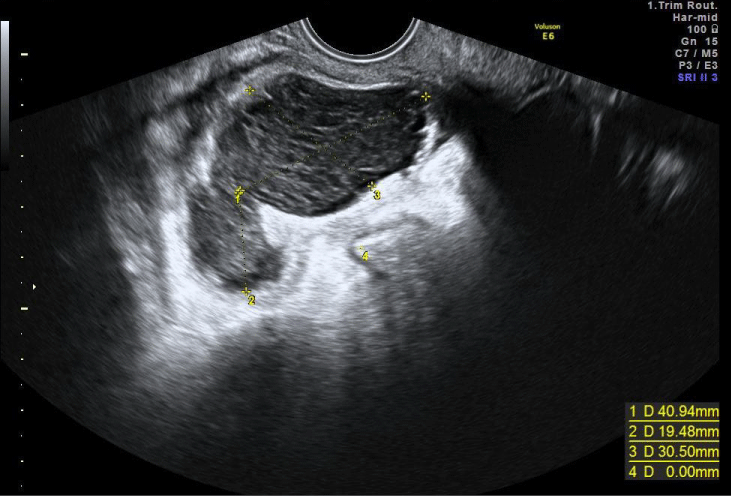 Figure 1: Transvaginal ultrasound scan at admission showing adnexal heterogeneous mass suggestive of tubo-ovarian abscess.
View Figure 1
Figure 1: Transvaginal ultrasound scan at admission showing adnexal heterogeneous mass suggestive of tubo-ovarian abscess.
View Figure 1
The patient was admitted to our inpatient Obstetrics ward.
She was medicated with intravenous (IV) Ceftriaxone 1 g every 12 hours, Erythromycin 500 mg every 6 hours and Clindamycin 900 mg every 8 hours, Ketorolac 30 mg and Paracetamol 1 g as needed.
During the stay at the hospital there was full clinical and analytic recovery. 48 hours after admission there was a slight clinical recovery, serum analysis revealed improvement of the inflammatory parameters (Leucocytes: 11.3 × 103/uL; CRP: 42.7 mg/L) and increase in beta hCG (312 mUI/mL), compatible with normal evolution of an intrauterine pregnancy. Ultrasound scan findings were similar to the admission; there was still no image of gestation sac inside the uterus. Serologies to Hepatitis B, C and HIV came negative.
Since then, the patient status kept on slowly but progressively improving and beta hCG levels suggested intrauterine viable pregnancy.
She completed 14 days of intravenous antibiotics.
At discharge, the patient was fully clinically recovered, with normal inflammatory values. At ultrasound, there was the same heterogeneous right adnexal mass with four cm of largest diameter (Figure 2) and no fluid at the Douglas Pouch and an intrauterine gestational sac with yolk sac (Figure 3).
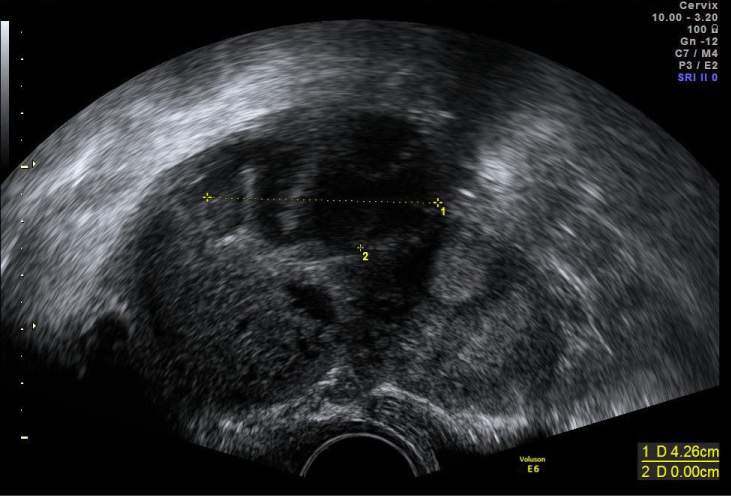 Figure 2: Transvaginal ultrasound scan showing tubo-ovarian abscess after 14 days of treatment.
View Figure 2
Figure 2: Transvaginal ultrasound scan showing tubo-ovarian abscess after 14 days of treatment.
View Figure 2
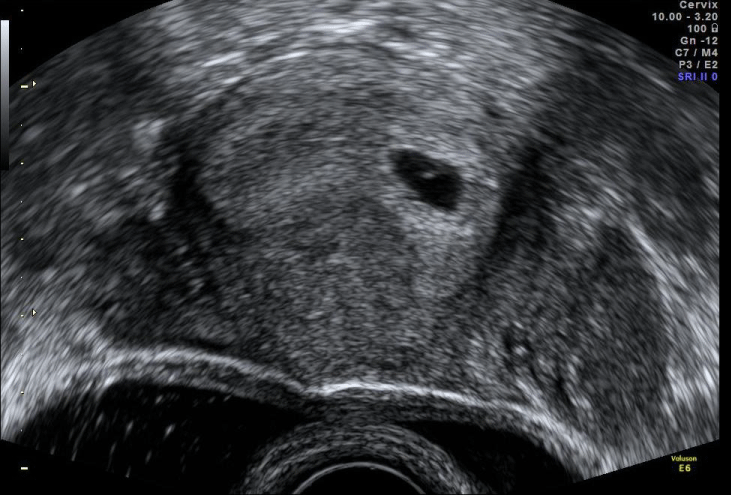 Figure 3: Intrauterine gestational sac with yolk sac inside.
View Figure 3
Figure 3: Intrauterine gestational sac with yolk sac inside.
View Figure 3
The patient was sent to our outpatient clinic but missed the appointments.
At the 10th week of gestation she returned to our emergency department with moderate/heavy vaginal blood loss. At ultrasound, there was a heterogeneous content of 17 mm of thickness with no image of gestational sac inside the uterine cavity. The same heterogeneous mass was present at the right adnexa, with similar size and characteristics of the one at discharge. The patient was submitted to uterine curettage by suction, after receiving 100 mg of Doxycycline per os (PO).
After confirming the uterine cavity was empty and the blood loss was controlled, the patient was discharged and medicated with 200 mg of Doxycycline PO. She was sent to our Gynaecology outpatient clinic to complete follow-up of the adnexal pathology but missed the appointments once again.
The main symptom of our patient was pelvic pain. She had a positive beta hCG and an empty uterine cavity with an adnexal mass, which raised the suspicion of ectopic pregnancy. There was also elevation of the inflammatory parameters and the clinical and ultrasound findings were more suggestive of tubo-ovarian abscess, so she was admitted with the diagnosis of PID with pregnancy of unknown location to receive IV antibiotic treatment and to rule out ectopic pregnancy. There was a slow, but progressive and clear clinical improvement and beta hCG levels kept on rising with values of an intrauterine viable pregnancy. At discharge there was indeed an intrauterine gestational sac with yolk sac, confirming the location of pregnancy. In the end, the clinical diagnosis was TOA during early pregnancy.
In general lines, PID treatment must include effective antibiotics against N. gonorrhoeae (e.g. cephalosporins), C. trachomatis (e.g.: tetracyclines, macrolides) and eventually anaerobes (e.g. Metronidazol or Clindamycin). The first line therapy recommended in non-complicated cases of PID is one bolus of intramuscular (IM) Ceftriaxone 250 mg and Doxycycline 100 mg twice a day PO during 14 days. Metronidazole 500 mg twice a day may be added also for 14 days, if TOA is suspected. Azithromycin 1 g PO once a week, two weeks may be an alternative to Doxycycline. When there is no clinical improvement or in complicated cases (i.e. immunosuppression, severe disease, large TOA), inpatient treatment with intravenous antibiotics is recommended with Cefoxitin 2 g IV every 6 hours and Doxycycline 100 mg PO twice a day or Clindamycin 900 mg IV every 8 hours with Gentamicin 1.5 mg/Kg IV every 8 hours followed by 14 days of Doxycycline 100 mg PO twice a day or Clindamycin 450 mg PO every 6 hours [2,6].
The use of some antibiotics is limited during pregnancy. Most of them are classified as class B or C by the FDA. Ceftriaxone (Class B) remains the cephalosporin of choice during pregnancy, in particular to treat N. gonorrhoeae infections. In general, cephalosporins are considered safe in pregnancy. Azithromycin and Clindamycin are both class B and considered adequate to use in pregnancy. Metronidazole is class B, it must be used with caution during the first trimester and its repeated use should be avoided, even though CDC clearly recommends its use during the whole pregnancy [7-9].
There is quite few information about which medication should be used to treat PID during pregnancy, due to the rarity of these entity during this period [7]. One of the suggested regimens is to use Cefoxitin 2 g three times daily plus erythromycin 50 mg/kg IV perfusion, with the possible addition of Metronidazole 500 mg IV three times daily. A similar regimen was chosen to treat our patient, with Ceftriaxone as alternative to Cefoxitin and Clindamycin in alternative to Metronidazol, and it was effective to treat the disease. Due to the slow clinical improvement, the pregnant status and the presence of an abscess, it was necessary to keep 14 days of intravenous therapy. After completing the antibiotics course, there was a persistent adnexal mass, although smaller. There is no consensus about the benefit of treating it surgically during the first trimester, so we opted for an expectant management and ultrasound imaging control, to which the patient did not attend.
As described in the literature, this case had a pour obstetric outcome, with partial resolution of the TOA and miscarriage.
Although extremely rare, pelvic inflammatory disease may appear in pregnant patients and it is associated with poor obstetric outcomes. In a female patient with low abdominal pain, positive beta hCG levels with empty uterine cavity and an adnexal mass, ectopic pregnancy must be ruled out, but other aetiologies must always be kept in mind. Both tubo-ovarian abscess and its treatment may be harmful to an ongoing pregnancy. Due to the paucity of data, there is still no consensus on which antibiotics to use in pregnant women with pelvic inflammatory disease, so treatment must be individualized, trying to use efficient medication with the least potential teratogenicity. It is still not known if surgery is indicated in pregnant patients with tubo-ovarian abscesses, especially during the first trimester.
The authors report no conflict of interests.