In this report, we present a challenging case of pedunculated left atrial myxoma which was initially diagnosed on Transthoracic Echocardiography (TTE) at 39 weeks and 2 days gestation in a grand-multigravida woman with previous normal deliveries. She was delivered by urgent caesarean section with resection of the myxoma on the following day. She had an unremarkable recovery. Physicians should be vigilant to any new onset maternal heart murmurs during pregnancy, and have a low threshold to screen with transthoracic echocardiography. A multidisciplinary team approach from anesthetists, cardiologists, cardiothoracic surgeons and maternal-fetal medicine specialists is essential to optimize fetal and maternal outcomes.
Atrial myxoma, Pregnancy, Cardiac tumour, Echocardiography
A 36-year-old indigenous female with gravida 7 para 6 was transferred at 39 weeks and 2 days' gestation with suspected maternal left atrial myxoma on Transthoracic Echocardiography (TTE) for further management. TTE was performed to investigate her recent symptoms of dyspnoea and auscultatory finding of grade 2 systolic murmur best heard in the mitral area. There were no additional symptoms or signs to suggest cardiac flow obstruction or systemic embolization. She was otherwise a healthy individual with no previous rheumatic heart disease or family history of cardiac disease. Her past obstetric history was relevant for six uncomplicated term vaginal deliveries. Thus far she had an uneventful pregnancy, and her only significant finding was BMI of 35 and iron deficiency anemia.
On admission, the patient was asymptomatic and vital signs revealed blood pressure of 135/80 mmHg, heart rate of 87 bpm, and oxygen saturation of 98% on room air. Laboratory analysis was unremarkable, except for hemoglobin level of 98 g/L. Electrocardiogram showed normal sinus rhythm. TTE confirmed a 4 × 1 cm mobile echo-dense mass arising from interatrial septum (LA side), and prolapsing through the mitral valve during diastole (Figure 1a and Figure 1b). The left ventricular ejection fraction (67%) and valvular function were normal. Non-stress test confirmed a reassuring fetal status in a cephalic presentation. Following discussion between cardiologists, cardiothoracic surgeons, obstetricians and anaesthetist, consensus decision was made to perform caesarean section first, with a view to proceed with cardiac surgery the following day after stabilizing the patient. She underwent an uncomplicated caesarean section with tubal ligation under general anesthesia, combined with Transesophageal Echocardiography (TEE) (Figure 2), and gave birth to a male infant, weighing 3.670 g with Apgar scores of 8 and 9 at 1 and 5 minutes, respectively. Hemi-sternotomy was employed and 30,000 Units of IV heparin was administered prior to her heart put on Cardiopulmonary Bypass (CPB) machine. A left atrial mass was resected from the interatrial septum and the defect in the septum was approximated with a patch. The CPB time was 59 minutes. The histology was consistent with a benign cardiac myxoma (Figure 3). She had an unremarkable recovery, and was discharged on day five post cardiac surgery. Short and long-term follow-ups are expected, as per cardiology clinic protocol.
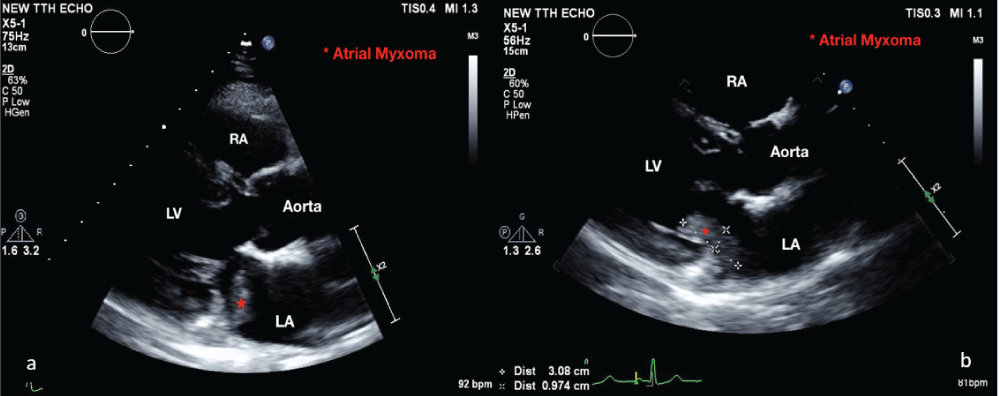 Figure 1: (a) Transthoracic echocardiography demonstrating an atrial myxoma in the left atrium during systole; (b) and protruding into the left ventricle during diastole (the red asterisk indicates atrial myxoma). View Figure 1
Figure 1: (a) Transthoracic echocardiography demonstrating an atrial myxoma in the left atrium during systole; (b) and protruding into the left ventricle during diastole (the red asterisk indicates atrial myxoma). View Figure 1
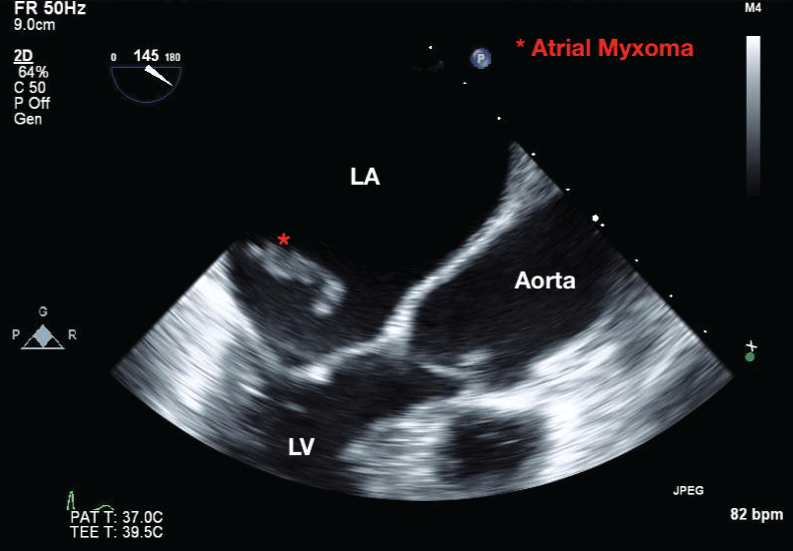 Figure 2: Transesophageal echocardiography demonstrating an atrial myxoma in the left atrium during systole (red asterisk). View Figure 2
Figure 2: Transesophageal echocardiography demonstrating an atrial myxoma in the left atrium during systole (red asterisk). View Figure 2
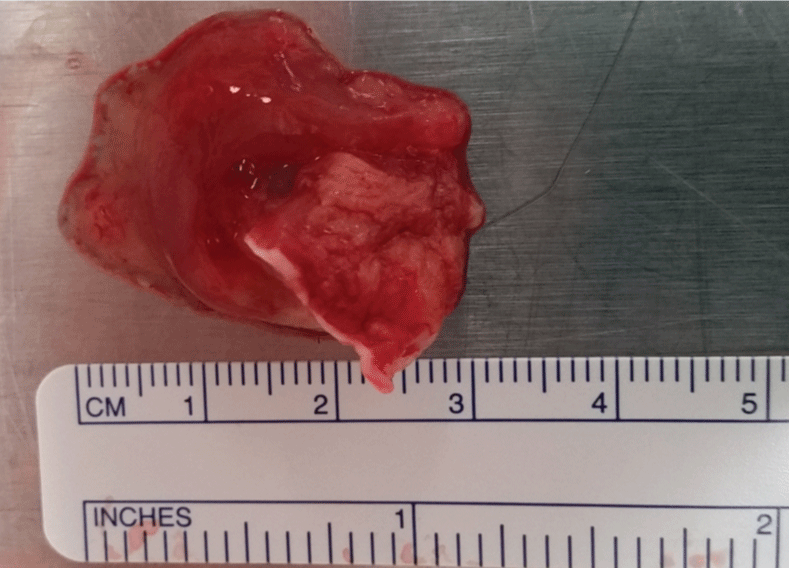 Figure 3: Histopathology examination confirming benign atrial myxoma. View Figure 3
Figure 3: Histopathology examination confirming benign atrial myxoma. View Figure 3
Atrial myxoma represents the most common benign tumor of the heart and typically (80%) originates from left atrium. Physical examination may reveal systolic or diastolic murmurs, depending on the location, size, and mobility of the myxoma. Clinical features range from asymptomatic cases to valvular obstructive manifestations, signs of systemic embolization, and various non-specific constitutional symptoms [1,2]. Pregnancy, being a hypercoagulable state, may further increase the risk of embolization. Once a diagnosis of myxoma has been suspected based on echocardiography, prompt resection, using cardiopulmonary bypass, is required due to the unpredictable risk of embolization, mitral inflow obstruction or cardiovascular events, including sudden death [3]. Histopathological examination is required to confirm the diagnosis. Recurrence is rare, but patients should have a long-term follow-up [4].
In this report, we present a challenging case of pedunculated left atrial myxoma which was initially diagnosed at 39 weeks and 2 days of gestation in a grand-multigravida woman with previous normal deliveries. Echocardiographic features of the mass being well-circumscribed, freely mobile from and not invading the surrounding structures suggest benign nature of the tumour. The appearance of the mass (globular, well-circumscribed, speckled), being pedunculated and its attachment site (interatrial septum on left atrial side) suggests it being a myxoma rather than atrial thrombus. Echocardiography can also help differentiating myxoma papillary fibroelastoma (normally small, round, attached to valve with 'pom-pom' appearance), rhabdomyoma (acoustic properties similar to myocardium, homogenous mass), fibroma (discrete, well demarcated echogenic mass within ventricular wall or septum) or malignant tumours (poorly defined, irregular shape, and inhomogeneous appearance). Normal cardiac structures such as pectinate muscles (small size, absence of independent mobility) and coumadin ridge (Q-tip appearance and normally seen between left atrial appendage and left upper pulmonary vein) were unlikely. Absence of the mass crossing tissue planes and being identified in multiple orthogonal views ruled out imaging artefacts whereas lack of inflammatory marker elevation and absence of signs of systemic illness makes infective vegetations unlikely.
The clinical dilemmas were the preferred mode of delivery (vaginal or caesarean), timing of cardiac surgery in relation to delivering the baby (one or two staged) and managing postpartum haemorrhage given need for high dose heparin in cardiac surgery. Given its unique situation, no consensus recommendation exists as to the preferred and the optimal mode of delivery, or for the timing of cardiac surgery in pregnant women with atrial myxoma. Considering the high and unpredictable risk of systemic embolization, myxoma resection should usually be performed with no delay. Obviously, cardiac surgery in early pregnancy has reduced maternal risk but may result in severe prematurity or fetal death in utero, mainly due to the effects of Cardiopulmonary Bypass (CPB). On the other hand, any delay in cardiac surgery may increase the risk of maternal sudden cardiac death. Strategies to reduce fetal risks associated with CPB should include; maintenance of uterine displacement, high flow rate (2.5 L/min per m2) and mean arterial pressure greater than 70 mmHg in order to maximize uteroplacental perfusion, using normothermic CPB and minimizing CPB times [5].
Recent review described the natural history of maternal cardiac myxomas during pregnancy over four decades, most however, were diagnosed either before or during the first, second, or early third trimester of pregnancy. Seven cases had early termination of pregnancy, and of the 26 reported deliveries with either caesarean section (n = 19) or vaginal delivery (n = 7), 20 (76.9%) were event-free survival, 4 (15.4%) were complicated, and 2 (7.7%) died. Neonatal outcome was not different between the modes of delivery or the timing of the cardiac surgery [6]. An earlier report from women undergoing cardiothoracic surgery during pregnancy also described the feasibility of seven cases having one-stage caesarean delivery to a healthy neonate (median gestational age 31 weeks) immediately prior to thoracotomy. However, none of the cases were due to myxoma, and none required anticoagulation dose to the same extent as usually given during myxoma resection [7].
In general, the following theoretical considerations should be made before deciding the mode of delivery; 1) It is expected to be the shortest in duration; 2) Maternal cardiac monitoring should be available to allow maternal heart rate adjustment (avoidance of tachycardia), especially during the intrapartum and postpartum hyperdynamic states; and 3) The delivery is anticipated to have the minimal risk of postpartum hemorrhage to allow the use of high-dose heparin during the cardiac surgery soon after. Given the patient's request for tubal ligation in our case, we felt that expediting delivery by caesarean section was justified and outweighed the risk of induced vaginal delivery. We feel that obtaining consent for Caesarean Hysterectomy for life-threatening events would be consistent with best practice.
In conclusion, we have described a diagnosis of left atrial myxoma in late pregnancy which was managed by urgent caesarean section and subsequent surgical resection via open heart surgery. Physicians should be vigilant to any new onset maternal heart murmurs during pregnancy, and have a low threshold to screen with echocardiography. Comprehensive multidisciplinary collaboration between thoracic anesthetists, cardiologists, cardiothoracic surgeons and maternal-fetal medicine specialists is essential to optimize fetal and maternal outcomes.