Among the most common chronic disorders of modern time, diabetes mellitus remains unique because of its multifactorial ramifications. Apart from the well-described association between congenital deafness and maternally inherited type of diabetes mellitus, the relationship between type 2 diabetes and hearing loss has been the subject of debate since Jordao reported a case of hearing loss with incipient diabetes coma almost 150 years ago. Documentation of this relationship among adult diabetics in developing countries (especially West Africa) where type 2 diabetes mellitus is prevalent has become imperative; hence this study.
1. To compare the hearing threshold levels among adult diabetes mellitus patients with non-diabetes mellitus subjects in south-western Nigeria.
2. To determine the type and severity of hearing loss among adult type 2 diabetic mellitus patients.
This was a 12 months prospective hospital based cross sectional study (December 2013- November 2014) of seventy consecutively selected adult diabetic patients and seventy age and sex matched non-diabetic control subjects.
The mean ages for the test and control groups were 48.4 years and 46.5 years respectively. Male to female ratio was 1:1. The Pure tone average as well as the hearing threshold levels for air and bone conduction was significantly higher for all frequencies tested except 0.25 KHz in the diabetic group compared to the non-diabetic control group.
Hearing threshold levels for adult diabetic patients are significantly higher than that of non-diabetic individual of a similar age.
Hearing threshold levels, Diabetes mellitus, Sensorineural, Pure tone audiometry
Hearing loss is the most frequent sensory deficit in human population, affecting more than 278 million people worldwide [1]. In the United States of America, it is estimated that hearing loss is twice as common in adults with diabetes compared to those who do not have the disease (prevalence of 21.3% in 4741 adults with diabetes mellitus, compared to 9.4% of the same population of adult without diabetes mellitus) [2].
The sensitivity of our hearing is indicated by the quietest sound that we can detect and this is regarded as the hearing threshold level [3]. Hearing threshold is defined as the lowest sound levels (absolute hearing threshold) that a listener can detect as well as the highest sound level that a listener can tolerate [4]. The thresholds are used to describe hearing sensitivity and the dynamic range of hearing of both normal and hearing impaired individuals [4]. The stimuli most frequently employed to obtain hearing thresholds and therefore used to measure hearing sensitivity are pure tones in the frequency range from 500 Hz-4000 Hz [5]. Though normal routine pure tone audiogram usually covers 8 frequencies in range of 125-8000 Hz.
Hearing threshold level (HL) in adult patients with type 2 diabetes mellitus has been described by some studies as a progressive, bilateral, sensorineural hearing loss of gradual onset that affects the low frequencies [6-9].
However, not all Researchers agree that type 2 diabetes mellitus (DM) can lead to sensorineural hearing loss (SNHL) or hearing loss at all; with some reporting a unilateral conductive or sensorineural pattern of hearing loss [9-11]. The hearing loss associated with DM maybe as a result of localized microangiopathy in the inner ear, neuronal degeneration or diabetic encephalopathy but might also be due to derangement in the metabolism of glucose and mechanism associated with hyperactivity of free oxygen radical [12,13].
These pathological changes and metabolic disturbance might result in a cochlear, retrocochlear, or combined cochlear-retrocochlear hearing disorder [14,15].
Auditory involvement in adult patients with type 2 DM have been reported to be either a gradual bilateral SNHL similar to presbycusis but with greater losses than expected for age range, a sudden onset of early SNHL affecting low frequencies or a low to mid frequency loss or even a unilateral and bilateral conductive hearing loss [16-20]. Some studies even describe DM as the possible cause of unilateral sudden hearing loss [21]. SNHL occurs when there is damage to the inner ear or to the nerves that travel from the ear to the brain [3].
These conflicting results regarding the hearing levels in diabetic patients were obtained from a population of both type 1 and type 2 diabetics. Since adult diabetics in south-western Nigeria are exclusively type 2 diabetics, it became necessary to undertake this study; which aims to determine the hearing threshold levels among adult diabetics in the south-western part of Nigeria.
This was a twelve months (December 2013 - November, 2014) prospective hospital based cross-sectional study of old and newly diagnosed adult patients with type 2 diabetes mellitus between the ages of 31 and 64 years presenting at the endocrinology clinic of Obafemi Awolowo University Teaching Hospitals complex, Ile-Ife, a tertiary institution covering over five states in the south-western region of Nigeria.
The non-diabetic control subjects for this study were recruited from all cadres of staff at the Obafemi Awolowo University Teaching Hospitals Complex, Ile-Ife.
Ethical clearance was sought and obtained for this work from the hospital Ethics review committee after which a written informed consent was obtained from individual participants of this study.
For patients with DM enrolled for the study (Group I):
1. Patients with biochemically and clinically diagnosed DM.
2. Patients between the ages of 31 - 64 years of age.
3. Patients who were in good mental health to give reliable answers to questionnaires.
4. Patients who were non-hypertensive.
Control group (Group II):
1. Non-diabetic subjects of ages between 31 - 64 years who had given consent for the study and are non-hypertensive.
For patients with DM enrolled for the study (Group I):
1. History of ear surgeries performed in the past.
2. History of infections in the ear, nose and throat in the past 3 months.
3. History of ototoxic drug use.
4. Patients who were 65 years of age and above.
5. Patients with occupational noise exposure.
6. Patients who were suffering from hypertension and chronic renal disease.
Control group (Group II):
Same as that for the study group except the control group does not have DM.
A measurement of the fasting blood sugar (FBS) was done in all the subjects using a calibrated Accu-Chek Active glucose meter machine (by Roche). The FBS of the control subjects was done in the morning shortly after the individual's arrival at the office for work. This was preceded by a prior announcement in these offices the day before. Those whose FBS value was less than 110 mg/dl (6.1 mmol/L), and had no clinical symptoms and signs suggestive of DM and also met other inclusion criteria were selected for the study.
Similarly the FBS for the diabetic subjects was done on the morning of their clinic visit, as it is customary for these patients to skip breakfast before a FBS is done in the clinic.
Diabetic patients had tuning fork examination done using a 512 Hz tuning fork. The essence and procedure for the clinical test was explained to these patients. The tuning fork was then struck gently against a bony prominence (the elbow) and placed behind the right ear (mastoid bone) and before the tone decays (bone conduction testing), the fork was brought to within 1 cm of the pinna, holding the prongs in a perpendicular position (air conduction testing). The same procedure was repeated for the left ear, and the results were noted. This was the RINNES test [19]. This test was positive when air conduction test was better than bone conduction test, and this was indicative of a normal hearing or sensorineural hearing loss (SNHL). A negative test was reported when the bone conduction testing was better than air conduction testing, this was indicative of a conductive hearing loss (CHL).
The tuning fork (512 Hz) was struck again gently against the elbow and placed on the glabella of the individual. Shortly after decay or after about 30 seconds, the tuning fork was removed and individual asked to reveal the ear (right or left) in which the sound was heard better. This was the WEBER'S test [22].
The same tuning fork tests were performed on the control group of patients with the same interpretations.
PTA was done for both group of participants in a sound proof booth using an ISO Standard calibrated clinical Audiometer (Interacoustic AD28). The air and bone conduction hearing levels were measured at different frequencies from 0.5 to 8.0 KHz with masking done for the bone threshold measurements. The pure tone hearing threshold average was calculated as the arithmetic mean for the air conduction tracings at four frequencies i.e. 0.5 KHz, 1 KHz, 2 KHz and 4 KHz.
Average hearing level for bone conduction was also calculated at similar frequencies and air- bone gap was calculated by subtracting the value of bone conduction average from that of air conduction average. Value of 15 dBHL and above was regarded as significant [22]. Thus a subject had CHL if air conduction average was increased and bone conduction average was within normal with or without a significant air-bone gap, Mixed hearing loss (MHL) if both air and bone conduction averages were increased but air conduction average showed an even greater increase and SNHL if both air and bone conduction averages were increased without a significant air-bone gap OR only bone conduction average is increased.
Low frequencies were between 0.25 KHz to ˂ 2 KHz and High frequencies were from 2 KHz and above. The slope of the PTA audiogram was noted for the type of frequency losses.
Based on the pure tone average in the better ear, hearing loss is classified into [1]:
→ Normal - 0 - 25 dBHL in the better ear
→ Mild - 26 - 40 dBHL (better ear)
→ Moderate - 41 - 60 dBHL (better ear)
→ Severe - 61 - 80 dBHL (better ear)
→ Profound - > 81 dBHLin the better ear.
The severity of the hearing loss for each subject was based on the WHO standard classification for the better ear.
Data obtained were recorded and analyzed using the statistical package for social sciences (SPSS) version 21. Frequency count and percentages were used for demographic variables. Multiple linear regression models were used for continuous variables and multinomial logistic regression models were used for categorical values. Level of statistical significance was set at p value 0.05.
Seventy adult patients with DM participated in this study and seventy healthy adult non-diabetic participants constituted the control group. There were 35 (50%) males and 35 (50%) females giving a male to female ratio of 1:1. The age range of the cases was 31 to 64 years with a mean age of 48.40 as shown in Table 1.
Table 1: Age and Sex Distribution of the study subjects and controls. View Table 1
Hearing loss was found in 71.4% (n = 50/70) of diabetic subjects compared to 21.5% (n = 15/70) of the non-diabetic control group as shown in Figure 1.
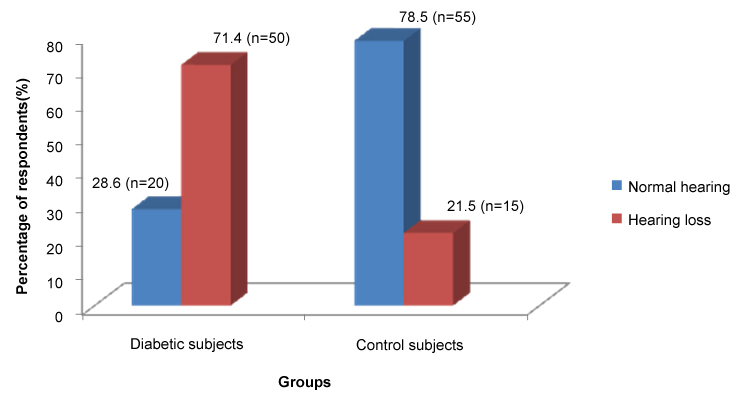 Figure 1: Bar chart showing the distribution of hearing loss in adult diabetic patients and controls. View Figure 1
Figure 1: Bar chart showing the distribution of hearing loss in adult diabetic patients and controls. View Figure 1
When the mean air and bone conduction levels in the better ear of diabetic and control subjects were compared, there was a significant difference. The diabetic group demonstrated an elevated hearing threshold level in all the frequencies evaluated (except 0.25 KHz). This was in contrast to the non-diabetic control group which demonstrated a normal hearing level in all frequencies tested as shown in Table 2 and Table 3.
Table 2: Comparison of Hearing threshold levels for air conduction in the better ear for diabetic subjects and non-diabetic control subjects (N = 140). View Table 2
Table 3: Comparison of Hearing threshold levels for bone conduction in the better ear for diabetic subjects and non-diabetic control subjects (N = 140). View Table 3
The PTA average for the diabetic test subjects was significantly higher than that of the control subjects (t-value = 6.23, p = 0.00) as shown in Table 4.
Table 4: Mean PTA air conduction average in adult patients with diabetes and control subjects. View Table 4
Demographic variables such as age, sex, social economic class and family history of DM did not affect the pure tone averages among Diabetic subjects as shown in Table 5.
Table 5: Showing the Multiple Regression Analysis of continuous PTA tracing of diabetic subjects with demographic variables. View Table 5
Sensorineural hearing loss (SNHL) was the most common type of hearing loss seen among 46 (65.7%) of the total 70 adult diabetic subjects in this study while conductive hearing loss (CHL) and SNHL were the most common types of hearing loss found in the 15 control subjects with hearing loss.
There was also no statistically significant association between types of hearing loss and each of the demographic variables as shown in Table 6 and Table 7.
Table 6: Univariate analysis of Types of Hearing loss with Age, sex, SES and family history of DM in Diabetic subjects (n = 70). View Table 6
Table 7: A summary of multinomial logistic regression analysis using Types of Hearing loss among Diabetics as outcome. View Table 7
Mild hearing loss was the most common form of severity seen among the 70 adult diabetic subjects in this study (n = 31, 44.3%). This was followed by a moderate hearing loss (n = 15, 21.4%) and severe hearing loss (n = 4, 5.7%). There was no statistically significant association between Severity of Hearing loss and the demographic variables. As shown in Table 8 and Table 9.
Table 8: Univariate analysis of severity of Hearing loss with Age, sex, SES and family history of DM in Diabetic subjects (n = 70). View Table 8
Table 9: A summary of multinomial logistic regression analysis using Hearing loss severity as outcome in Diabetic subjects. View Table 9
Of the 70 diabetic subjects in this study, 62 (89%) were on oral hypoglycemic drugs (OHD) alone while 5 (7%) were on oral hypoglycemic drugs and insulin injection therapy. A further 3 (4%) were on diet and exercise therapy. The severity of hearing loss was less among the group of diabetics on a combined therapy of oral hypoglycemic drugs and insulin injections than those on oral hypoglycemic drugs alone who in turn had a better hearing than those on diet and exercise therapy only as shown in Figure 2.
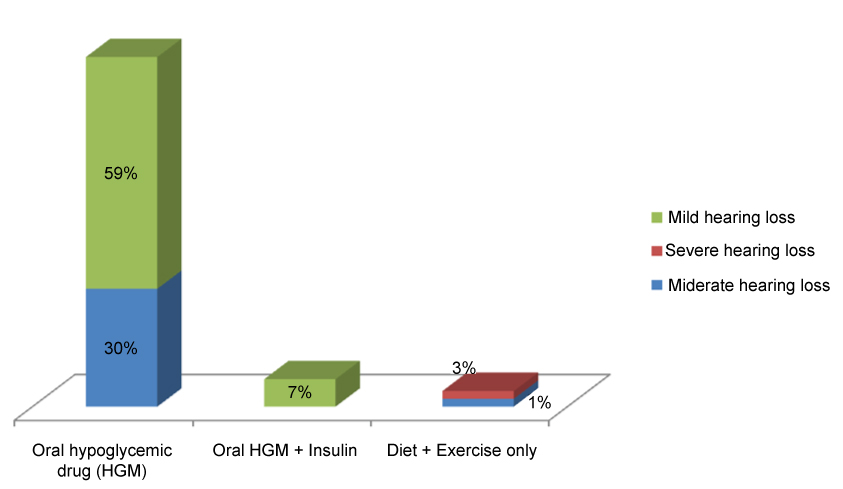 Figure 2: Bar chart showing the use of medications among diabetic subjects and their effect on the severity of hearing loss. View Figure 2
Figure 2: Bar chart showing the use of medications among diabetic subjects and their effect on the severity of hearing loss. View Figure 2
Opinions on the nature of the relationship between diabetes mellitus (DM) and an increased hearing threshold level indicative of hearing loss is still divergent despite many authors reporting a significant hearing loss in DM patients and others reporting the contrary. Among authors who reported significant hearing loss in diabetic patients, there are different views as to whether this relationship between diabetes mellitus and hearing loss is a causal one.
The mean ages in this study for cases and control subjects were 48.40 years and 46.53 years respectively. This is similar to the mean ages for test and control subjects reported by Ologe, et al. [23], who reported that individuals between 40-60 years of age with Type 2 diabetes were more likely to have hearing loss than their non-diabetic compatriots of similar age. Sakuta, et al. and Bainbridge, et al. also reported a statistically significant higher prevalence of hearing loss among middle aged diabetic subjects when compared to their compatriots of a similar age [2,24].
The prevalence of hearing loss among diabetics in the index study was 71.4%.When compared to the controls (21.6%) of the same age group it was statistically significant (p = 0.001). This finding was corroborated by Rajendran, et al. [25] reported a prevalence of 73% hearing loss in diabetic patients compared to 6.7% among non-diabetic control patients of the same age group. They worked on a similar population with similar inclusion and exclusion criteria as the present study. Afaf, et al. noted in general that the prevalence of hearing loss in the diabetic group was significantly higher than in the control group (p = 0.005) with no significant difference between gender in both groups with regards to the ear with better hearing [26].
Furthermore, a raised hearing level for air and bone conduction in all frequencies except 0.25 KHz was the predominant finding in 50 of the 70 diabetic subjects in this study (n = 50/70).When compared to the non-diabetic control subjects in this study it was statistically significant for all the frequencies except 0.25 KHz. This observation is in consonance with findings by Ologe, et al. working in Ilorin [23]. Cayönu, et al. working on elderly type 2 diabetic patients also reported significantly higher mean hearing levels across all the frequencies tested (except 0.25 KHz) in diabetic patients compared to non-diabetic controls [27]. Analysis of this observation and the very similar findings from this study points to the suggestion that age might not be a confounding factor for hearing loss in type 2 diabetic patients. Kakarlapudi, et al. found the effect of age on auditory thresholds in diabetic subjects to be clinically and statistically insignificant [28].
This study found the mean PTA average recorded for diabetic subjects were significantly higher than that of the non-diabetic control group (t = 6.23, p = 0.00). Dalton, et al. in a study of 344 adult patients with type 2 diabetes mellitus and defining the pure tone average as the mean hearing level over four frequencies (500, 1000, 2000 and 4000 Hz) as the present study, found a weak association between type 2 diabetes and hearing loss. Though he noted that the pure tone averages for type 2 diabetic patients were higher than that of the non-diabetic control group, the difference was statistically insignificant [29].
Sensorineural hearing loss was observed to be the most prevalent type of hearing loss among adult diabetic patients. This is in keeping with the predominant type of hearing loss among diabetic subjects described by Muhammad, et al. in a survey of 110 patients with diabetes mellitus reported SNHL in 87 (79%) of them [30]. Gazzaz, et al. in an observational retrospective study of 100 adult patients with DM reported SNHL as the most common type of otological disorder among diabetics [31].
A mild to moderate hearing loss was observed to be the most predominant forms of severity seen in our study (44.3%). Mozaffari, et al. in a survey of 71 non-elderly diabetic patients also reported mild to moderate hearing loss to be more common than severe hearing loss and that the severity of hearing loss increased with an increase in the duration of diabetes mellitus [32]. Muhammad, et al. found that a diabetic patient whose duration of diabetes mellitus was 5 years and less had less severity than those whose duration of diabetes was over 5 years [30]. This is in keeping with the findings of this study as majority of the participants were short term diabetics with mean duration of diabetes mellitus being only 5.2 years. This likely explains why mild to moderate hearing loss were the predominant pattern of severity found among patients in this study.
It has been reported that diabetic patients on medications especially injectable anti-diabetic drugs such as insulin have a less severe form of hearing loss [28]. Wackym, et al. observed that diabetic patients treated with diet and exercise alone had more severe hearing loss than those taking oral hypoglycemic agents who had worse hearing than the patients taking insulin [33]. This they attributed to the better metabolic control of the injectable medications [33]. This finding is corroborated by this study as diabetic patients who combined oral hypoglycemic drugs with injectable insulin had a milder form of hearing loss compared to the group of patients on diet and exercise alone and some of the patients on oral hypoglycemics only.
Hearing loss is more prevalent in diabetic patients than non-diabetic individuals. The Pure tone averages for adult diabetic patients are also significantly higher than that of non-diabetic individuals of a similar age. There is no gender predilection for hearing loss among individuals with diabetes mellitus.
Sensorineural hearing loss (SNHL) is the most common type of hearing loss among diabetic patients. Mild and moderate hearing losses are the predominant form of severity and there is no significant difference between male and female diabetic patients with regards to the type and severity of hearing loss.
Diabetic patients' especially middle aged individuals should have regular routine ear examinations and hearing tests. This is to aid early detection of hearing impairment as well as facilitate early clinical intervention with a consequent improvement in the quality of life of these patients. High frequency range audiometric testing should be considered for proper assessment of the level of impaired hearing in diabetic patients.
Further research on the subject of diabetes mellitus and hearing loss is advocated for, especially on the relationship between glyceamic control and severity of hearing loss.
Funding: None.
Conflict of interest: None declared.
Ethical approval: Obtained.