The present report describes the case of an 18-year-old male exhibiting Acute Lymphocytic Leukemia (ALL). He initially presented with a multitude of rheumatic manifestations that were attributed to Systemic Lupus Erythematosus (SLE). The initial peripheral smear and bone marrow exam did not show any abnormal cells that developed later on along his disease course.
Systemic lupus erythematosus, Acute lymphoblastic leukemia, Paraneoplastic rheumatic disorders, Lupus-like Syndrome, ANA
ACL: Anticardiolipin; ACR: American College of Rheumatology; ALL: Acute Lymphoblastic Leukemia; AML: Acute Myeloid Leukemia; ANA: Antinuclear Antibodies; Anti-dsDNA: Anti-Double Stranded DNA Antibodies; CML: Chronic Myeloid Leukemia; CMV: Cytomegalovirus; DIP: Distal Interphalangeal; EBV: Epstein Barr Virus; HBV: Hepatitis-B Virus; HCV: Hepatitis-C Virus; LA: Leukemic Arthritis; LCV: Leukocytoclastic Vasculitis; LDH: Lactate Dehydrogenase; MCP: Metacarpo-Phalangeal; PIP: Proximal Interphalangeal; RDs: Rheumatic Disorders; SLE: Systemic Lupus Erythematosus; SLICC: Systemic Lupus International Collaborating Clinics
Paraneoplastic Rheumatic Disorders (RDs) are referred to the rheumatic manifestations that are caused by a hidden malignant disease; however, they are not directly related to invasion by a tumor or its metastasis. The paraneoplastic RD usually precede the onset of the malignancy by months or years, and they improve once treatment is initiated for the underlying malignancy [1].
Acute Lymphoblastic Leukemia (ALL) is one of the hematological malignancies that frequently present with musculoskeletal symptoms [2]. Lupus-like syndrome has been described in association with various malignancies, including breast carcinoma, ovarian carcinoma and hairy-cell leukemia [3]. This report describes an 18-year-old male patient who developed arthritis, fever, pancytopenia and positive Antinuclear Antibodies (ANA). Repeated peripheral smear twice and bone marrow exam did not show any abnormal cells. Months later, a repeat bone marrow biopsy diagnosed acute lymphoblastic leukemia.
An 18-year-old Caucasian male presented with high fever, progressive non-itchy erythematous maculopapular skin rash and blackish crusty lesions involving the extremities, trunk and the external genitalia. Few months later the patient noticed violet discoloration around the eyes, hair loss, malar rash, and orogenital ulcers, as well as arthritis of both elbows and knees. He also developed loss of weight, subconjunctival hemorrhage & anemic manifestations. Shortly after, he suffered from puffiness of the eye lids associated with bilateral lower limb edema.
Examination revealed fever (39 °C), soft palate wet purpura, subconjunctival hemorrhage, heliotrope like rash, maculopapular, vesicular rash with some blackish crusty lesions and secondary infected areas involving his extremities, and to a lesser extent the trunk (Figure 1 and Figure 2). Some lesions were observed over the scrotum. There was also bilateral lower limb oedema together with bilateral tender and swollen elbows and knees.
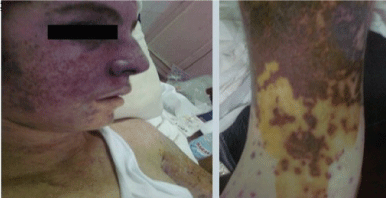 Figure 1: A) Macuolopapular rash with heliotrope rash over the face (left panel); (B) Macuolopapular rash over the arm (right panel). View Figure 1
Figure 1: A) Macuolopapular rash with heliotrope rash over the face (left panel); (B) Macuolopapular rash over the arm (right panel). View Figure 1
 Figure 2: Dusky red macular and bulbous bullous lesions seen over the lower limb. View Figure 2
Figure 2: Dusky red macular and bulbous bullous lesions seen over the lower limb. View Figure 2
Investigations revealed pancytopenia (HB: 5.4 g/dl, MCV: 84 fl, TLC: 2600/mm3, and PLT: 28000/mm3) with normal differential leukocytic count and no abnormal cells, reticulocytopenia, elevated ESR (55 mm/hr) and elevated Lactate Dehydrogenase (LDH). Bone marrow aspiration revealed a moderately hypercellular bone marrow. Urine analysis showed proteinuria with no casts; protein in 24 hours urine was 1125 mg/24 h. There was no hypocomplementemia. Blood culture and sensitivity showed no growth.
The autoimmune profile was only positive for homogenous Antinuclear Antibodies (ANA) at a titer of 1/80 by immune fluorescence (our hospital lab reference was 1/40). Virology screen for Hepatitis-C Virus (HCV), Hepatitis-B Virus (HBV), Epstein Barr Virus (EBV) and Cytomegalovirus (CMV) were negative. Echocardiograhy, chest X-ray & abdominal ultrasonography were normal. Skin biopsy showed vascular and perivascular infiltration of polymorphonuclear leukocytes with formation of nuclear dust, extravasation of erythrocytes, and fibrinoid necrosis of the vessel walls; a picture consistent with leukocytoclastic vasculitis.
The multisystem nature of the disease and the rheumatic manifestations (malar rash, oral ulcers, arthritis, proteinuria, leukopenia, thrombocytopenia, positive ANA above the laboratory reference range) were in favor of Systemic Lupus Erythematosus (SLE) as they met the 2012 Systemic Lupus International Collaborating Clinics (SLICC) classification criteria for SLE [4].
Intravenous methyl-prednisolone 3-gram over three days was started. The patient was maintained on prednisolone 40 mg/day together with hydroxychloroquine 400-mg/day, gastric protection and antiresorptive therapy. The patient was discharged with marked improvement of fever, skin lesions (Figure 3), general condition and all the initially abnormal laboratory investigations. He was scheduled for a follow-up visit after one month.
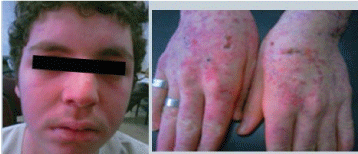 Figure 3: Patient's pictures on discharge showing marked improvement of all skin lesions. View Figure 3
Figure 3: Patient's pictures on discharge showing marked improvement of all skin lesions. View Figure 3
One month later the patient was readmitted to the hospital with high grade fever and extensive skin crusty bullous lesions over knees and legs (Figure 4). Investigations revealed pancytopenia with relative lymphocytosis and some atypical lymphocytes. BM aspiration and biopsy showed all elements to be depressed and the bone marrow to be totally infiltrated by 70% of blast cells. Blasts were myeloperoxidase stain negative, features suggestive of Acute Lymphoblastic Leukemia (ALL). The patient was transferred to The National Cancer Institute and missed his follow up.
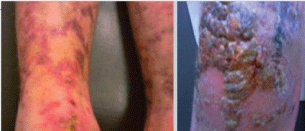 Figure 4: Multiple bulbous bullous-like lesions with crusty areas over knee and livedo like lesions over legs. View Figure 4
Figure 4: Multiple bulbous bullous-like lesions with crusty areas over knee and livedo like lesions over legs. View Figure 4
Paraneoplastic syndromes are the manifestations of an underlying malignancy that may be hidden. The paraneoplastic rheumatic disorders include a wide spectrum of manifestations for example; arthritis, myositis, lupus-like syndrome and vasculitis. They may be an initial presentation, in such cases the diagnosis may be difficult and needs a high index of suspicion. However they can be used as a clue for the underlying malignancy [5].
Paraneoplastic RD can be mediated by the following assumed mechanisms: (i) A common initiative factor (bacteria, virus, radiation) that can trigger both the paraneoplastic RD and the malignancy, (ii) Substances released by tumor cells, for example hormones and peptides, which trigger inflammation and (iii) Hypersensitivity reaction, which develops against the released intracellular antigens from apoptotic tumor cells, including nucleic acid-associated proteins; the latter explanation is supported by the presence of auto-antibodies to nuclear proteins in patients with paraneoplastic RDs [6].
In our case the misleading diagnosis was influenced by the presence of Leukocytoclastic Vasculitis (LCV), arthritis, proteinuria and blood abnormalities together with positive ANA. Cutaneous Leukocytoclastic Vasculitis is the most common type of vasculitis reported with cancer, especially the myeloproliferative and lymphoproliferative hematologic malignancies [7]. Fain, et al. evaluated 60 patients with vasculitis and malignancy, LCV was encountered in 45% of cases in their cohort [8].
Also Leukemic Arthritis (LA) has been reported more frequently in children, and in acute leukemia more than chronic subtypes. Synovial infiltration by leukemic cells resulting in hemorrhage into the joint appears to be the primary potential pathogenic mechanism. Other mechanisms include immune complex-induced synovitis and synovial reaction to periosteal or capsular infiltration [9]. Asymmetrical oligoarticular or polyarticular arthritis of sudden onset, with predominance of the large peripheral joints of lower extremities is usually encountered. Rheumatoid factor is frequently negative and radiographs tend to be normal [1].
On the other hand, the typical presentation of lupus arthritis is a symmetric polyarthritis that tends to involve the small joints more than the large ones although any joint may be affected. The hand joints including the Metacarpophalangeal (MCP), Proximal Interphalangeal (PIP) and Distal Interphalangeal (DIP) are commonly involved. Shoulders, ankles and elbows are less commonly affected. The isolated involvement of a large joint should raise the possibility of an alternative explanation other than lupus-related arthritis [10].
Again glomerular disease has been reported in all hematologic malignancies. The list of presentations includes nephrotic syndrome, subnephrotic-range proteinuria, haematuria, and overt renal failure. In ALL minimal changes and focal segmental diseases are the common glomerular lesions Diagnosis of glomerulopathy can often precede a diagnosis of a haematological malignancy [11].
Finally Anti-Nuclear Antibodies (ANA) have been demonstrated in the sera of patients with malignant diseases mainly those with solid tumors and lymphoproliferative disorders. Solans-Laque, et al. found a significant prevalence of ANAs (27%) among cancer patients with different etiologies compared to other auto-antibodies. Low ANA titers (1:80) were most often detected. 17.1% of their cohort patients with positive ANAs developed paraneoplastic RD [12]. A higher titer of ANA (1/640) was reported in a 22-year-old male patient who was found to have ALL. The patient presented initially with joint and muscle aches, pleurisy, fever, weight loss, hyperuricemia, leukopenia, and mild thrombocytopenia, although repeated peripheral smears did not show any abnormal cells. The persistent weight loss, in spite of steroid treatment of the presumed SLE, alerted the author to the possibility of a hematological malignancy. A diagnosis of ALL was confirmed by bone marrow biopsy 6 months after his initial presentation [13].
In general ALL is not associated with autoantibody formation. Presence of ANA in children with leukemia has occasionally been reported [2]. However its clinical significance is not known, it may reflect a breakdown in self tolerance associated with cancer that leads to an antigen-driven immune reaction [14]. An abnormal titer of ANA by immunofluorescence or an equivalent assay has been established as one of the diagnostic serologic markers for SLE, as outlined by both the revised 1997 American College of Rheumatology (ACR) [15] and the new SLICC classification criteria [4]. However ANA at a titer of ≥ 1:160 is generally accepted as a significant one, because it is less commonly encountered in healthy individuals [16].
Anti-double stranded DNA antibodies (anti-dsDNA) are more specific to SLE [16], and are rarely encountered in hematological malignancies. Among 129 patients with different hematologic malignancies [including Acute Myeloid Leukemia (AML), Chronic Myeloid Leukemia (CML), ALL and lymphoma], as well as other malignant diseases who were examined by a radio immunological technique, none of them exhibited a positive reaction for anti-dsDNA [17].
The findings in our patient illustrate that ALL in adolescents may mimic SLE. Male sex, atypical arthritis, elevated Lactate Dehydrogenase (LDH) that could not be accounted for (mainly by hemolysis) together with the presence of positive ANA at low titer should alert the physician to the possibility of malignancy. In such a setting a positive ANA should be interpreted with caution particularly when no other SLE specific antibodies (anti dsDNA & anti Sm) can be elicited.
The Rheumatology & Clinical Immunology Unit, Internal Medicine Department, Faculty of Medicine, Cairo University, Egypt.
None to declare.