Central pancreatectomy is performed to preserve pancreatic function in selected patients with low-grade tumors. We evaluated short-term and long-term pancreatic function and nutritional status after central or distal pancreatectomy.
The subjects were 24 patients undergoing central pancreatectomy and 91 patients receiving distal pancreatectomy. We retrospectively evaluated body weight, serum albumin, hemoglobin A1c, and complications.
After central pancreatectomy, body weight and hemoglobin A1c did not change significantly up to 60 months postoperatively compared with before surgery, while serum albumin was significantly increased at all postoperative assessments (6, 12, 36, and 60 months, all P < 0.05). After distal pancreatectomy, body weight did not change significantly at any time, while serum albumin was significantly increased until 36 months (all P < 0.001), and hemoglobin A1c was significantly increased at all postoperative assessments (all P < 0.0001). Following distal pancreatectomy, fatty liver occurred in 11 patients (12%), 43 patients (47%) required pancreatic digestive enzymes, and 20 patients (30%) developed diabetes, while none of these complications occurred after central pancreatectomy (P = 0.0733, P = 0.0001, and P = 0.0024, respectively). After central pancreatectomy, the incidence rate of post-operative pancreatic fistula Grade B + C was 24%, while the incidence rate of postoperative pancreatic Grade B + C was 11% (P = 0.0947).
Compared with distal pancreatectomy, central pancreatectomy preserves both short-term and long-term pancreatic function and nutritional status.
Central pancreatectomy, Distal pancreatectomy, Nutrition, Diabetes mellitus
The pancreas has both exocrine and endocrine functions, and plays an important role in digestion and absorption by secreting pancreatic digestive enzymes in the pancreatic juice and releasing insulin into the blood. In recent years, central pancreatectomy has been performed to preserve pancreatic function in selected patients with low-grade pancreatic tumors, but its short-term and long-term efficacy for achieving this objective has been not so clear.
Theoretically, pancreatic exocrine function should be better after central pancreatectomy than after other types of pancreatectomy because more of the pancreas is retained. Because patients with low-grade pancreatic tumors can be expected to have longer survival after central pancreatectomy, preservation of pancreatic function over the long-term is required to maintain an acceptable quality of life.
Pancreatogenic diabetes, which is classified as type 3c by the American Diabetes Association [1], is associated with various diseases or conditions such as pancreatitis, benign and malignant tumors, cystic fibrosis, hemochromatosis, fibrocalculous pancreatopathy, trauma, and pancreatectomy. In Western countries, 8-9% of all patients with diabetes have type 3c disease and 2-3% developed diabetes after pancreatectomy [2,3]. Accordingly, the importance of also maintaining endocrine pancreatic function after pancreatectomy has been emphasized.
We conducted the present study to evaluate the short-term and long-term changes of pancreatic function and nutritional status after central pancreatectomy and performed a comparison with the outcome after distal pancreatectomy.
The subjects were 115 patients who underwent central pancreatectomy (n = 24) or distal pancreatectomy (n = 91) at our department (Department of Surgery, Tokyo Women's Medical University, Tokyo, Japan) from January 2005 to December 2015 for whom complete data were available, and who had no residual pancreatic tumor, no other cancer, no other diseases that could influence their nutritional status. Retrospective evaluation of body weight, serum albumin, and hemoglobin A1c (HbA1c) before pancreatectomy and at 6 months, 12 months, 36 months, and 60 months after pancreatectomy was performed. Postoperative occurrence of fatty liver, pancreatic exocrine failure (requirement for pancreatic digestive enzymes), and pancreatic endocrine failure (diabetes) was also evaluated. And we evaluated the incidence rate of postoperative pancreatic fistula according to the international study group of pancreatic fistula [4]. Data on the preoperative and postoperative status were obtained from the medical records. This study was approved by the ethical committee of our hospital (Figure 1).
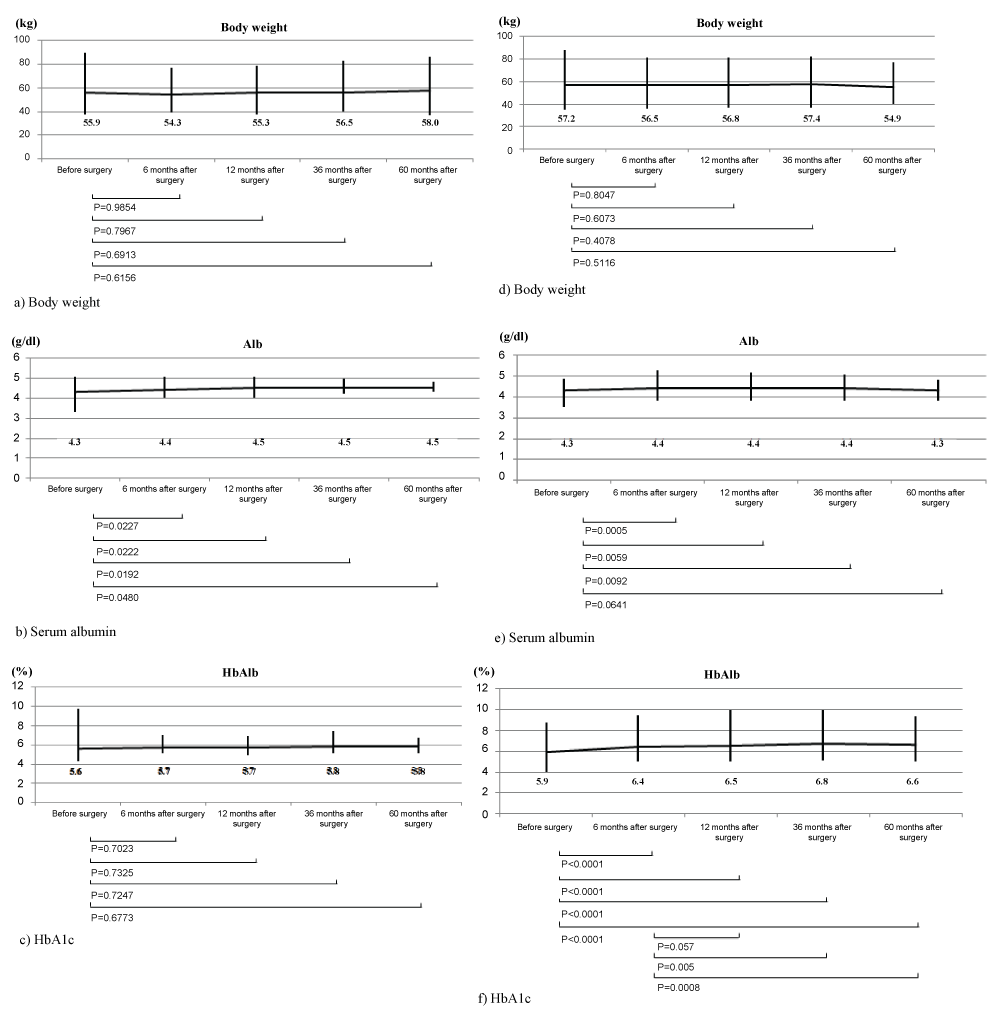 Figure 1: Parameters before and 3, 6, 12, 36, and 60 months after surgery. Data were analyzed using the t-test.
View Figure 1
Figure 1: Parameters before and 3, 6, 12, 36, and 60 months after surgery. Data were analyzed using the t-test.
View Figure 1
When the tumor was located in the pancreatic body and it was easy to separate the tumor from the splenic artery and vein and to perform an anastomosis between the pancreatic tail remnant and small intestine, we selected central pancreatectomy. Also, when the diameter of the pancreatic remnant was more than 3 cm, we tried central pancreatectomy. When the tumor was located in the pancreatic tail or in the pancreatic body and it was difficult to separate the splenic artery and vein, we selected distal pancreatectomy. Pancreatic body and tail were defined as by the General Rules for the Study of Pancreatic Cancer published by Japan Pancreatic Society [5].
Preoperative values were defined as the most recent values obtained within one week before pancreatectomy, while postoperative values were obtained at each evaluation time point or within one month before or after it. Fatty liver was defined as being present if the CT value of the liver was ≤ 50 HU on plain CT scans, or the liver/spleen attenuation ratio (CT value of the liver/CT value of the spleen) was < 0.8. After the operation, patients underwent CT or US every 6-12 months.
Replacement of pancreatic digestive enzymes was performed in patients who had persistent diarrhea with associated weight loss and low serum albumin levels, as well as in patients who were considered to be at risk of such outcomes. We used pancrelipase or excelase as pancreatic digestive enzyme.
HbA1c was expressed as National Glycohemoglobin Standardization Program (NGSP) values, and all HbA1c levels calculated according to the Japan Diabetes Society (JDS) method were converted to NGSP values by the following formula: NGSP HbA1c (%) = JDS HbA1c (%) + 0.4%. Diabetes mellitus was defined as being present if HbA1c (NGSP) was > 6.5%.
Results are expressed as numbers (percentages) or as mean values (with ranges), except that age is expressed as the median (range). Wilcoxon’s rank sum test was used to compare ages, the chi-square test was employed to compare categorical variables, and Student’s t-test was performed to compare continuous variables. A probability value of less than 0.05 was considered to indicate statistical significance. All analyses were performed using JMP Pro ver. 11.2 software.
The central pancreatectomy group comprised 5 men and 19 women with a median age of 54 (range: 31-70) years, mean height of 158.8 (140-175) cm, mean weight of 55.9 (38-89.3) kg, mean body mass index of 22.1 (16.2-31.8) kg/m2, mean serum albumin of 4.3 (3.3-5.1) g/dl, and mean HbA1c of 5.6 (4.3-9.8)%. Primary diseases were Intraductal Papillary Mucinous Neoplasm (IPMN) in nine patients (38%), Neuroendocrine Tumor (NET) in nine patients (38%), Solid Pseudopapillary Neoplasm (SPN) in three patients (13%), and Mucinous Cystic Neoplasm (MCN), pancreatic cancer, and acinar cell adenoma in one patient (4%) each. The mean observation period was 5.64 (1.83-11.03) years.
In the distal pancreatectomy group, there were 31 men and 60 women with a median age of 57 (19-78) years, mean height of 159.6 (138.4-180) cm, mean weight of 57.2 (35.2-88.1) kg, mean body mass index of 22.3 (15.0-33.1) kg/m2, serum albumin of 4.3 (3.5-4.9) g/dl, and HbA1c of 5.9 (4.0-8.7)%. Primary diseases were IPMN in 42 patients (46%), NET in 20 patients (22%), SPN in 13 patients (14%), MCN in 14 patients (15%), and pancreatic cancer and serous cyst adenoma in one patient (1%) each. The number of pancreatic cancer was very low, because other pancreatic cancer's patients were recurrence or underwent adjuvant chemotherapy. So these cases were excluded in this study. The mean observation period was 4.96 (1.08-10.68) years.
In both groups, there were no patients with preoperative fatty liver.
There were no significant differences of preoperative characteristics between the central pancreatectomy group and the distal pancreatectomy group (Table 1).
Table1: Clinical characteristics. View Table 1
Data was available for all patients before pancreatectomy and at 6 and 12 months. For 88 patients’s data was available at 36 months and for 52 patients’s data was available for all time points, including at 60 months.
At 6 months, 12 months, 36 months, and 60 months after central pancreatectomy, compared with the preoperative values, body weight was respectively 54.3 kg (P = 0.9854), 55.3 kg (P = 0.7967), 56.5 kg (P = 0.6913), and 58.0 kg (P = 0.6156), serum albumin was respectively 4.4 g/dl (P = 0.0227), 4.5 g/dl (P = 0.0222), 4.5 g/dl (P = 0.0192), and 4.5 g/dl (P = 0.048), while HbA1c was respectively 5.7% (P = 0.7023), 5.7% (P = 0.7325), 5.8% (P = 0.7247), and 5.8% (P = 0.6773) (Table 2: a-c). There were no significant short-term or long-term differences of the body weight or HbA1c compared with the preoperative values, but albumin levels were significantly increased from 6 months after central pancreatectomy.
Table 2: Postoperative fatty liver. View Table 2
In the distal pancreatectomy group, body weight was respectively 56.5 kg (P = 0.8047), 56.8 kg (P = 0.6073), 57.4 kg (P = 0.4078), and 54.9 kg (P = 0.5116), serum albumin was respectively 4.4 g/dl (P = 0.0005), 4.4 g/dl (P = 0.0059), 4.4 g/dl (P = 0.0092), and 4.3 g/dl (P = 0.0641), and HbA1c was respectively 6.4% (P < 0.0001), 6.5% (P < 0.0001), 6.8% (P < 0.0001), and 6.6% (P < 0.0001) (Table 2: d-f). There were no significant short-term or long-term differences of body weight compared with the preoperative value. Albumin levels were significantly increased from 6 to 36 months after distal pancreatectomy, but no significant difference was observed at 60 months. HbA1c was significantly higher at all times after distal pancreatectomy.
In the central pancreatectomy group, none of the patients developed fatty liver, required replacement of pancreatic digestive enzymes, or developed diabetes after pancreatectomy. In contrast, 11 patients (12%) developed fatty liver in the distal pancreatectomy group; while 43 patients (47%) required replacement of pancreatic digestive enzymes and 20 patients (30%) developed diabetes. The incidence of requirement of pancreatic digestive enzymes and the incidence of developing diabetes were significantly higher in the distal pancreatectomy group, but there was no significant difference in the incidence of fatty liver (P = 0.0001, P = 0.0024, and P = 0.0733, respectively) (Table 3 and Table 4). After distal pancreatectomy the incidence rate of the postoperative pancreatic fistula Grade B + C was 11% (10 patients), while after central pancreatectomy the incidence rate of the postoperative pancreatic fistula Grade B + C was 24% (6 patients). There was no significant difference (P = 0.947).
Table 3: Postoperative need for pancreatic enzyme supplementation. View Table 3
Table 4: Preoperative and postoperative diabetes mellitus. View Table 4
The main findings of the present study were as follows: 1) After central pancreatectomy, body weight and HbA1c did not change significantly up to 60 months postoperatively, while serum albumin was significantly increased at all times. 2) After distal pancreatectomy, body weight did not change significantly, while serum albumin was significantly increased until 36 months and HbA1c was significantly increased at all times. 3) Fatty liver, requirement for pancreatic digestive enzymes, and new-onset diabetes were all frequent following distal pancreatectomy, while none of these complications occurred after central pancreatectomy.
These findings suggest that central pancreatectomy better preserves both short-term and long-term pancreatic function and nutritional status, especially pancreatic function.
When pancreatectomy is performed for pancreatic tumors, the two important considerations are curability by surgical resection and preservation of pancreatic function. The pancreas has both exocrine and endocrine functions, and plays an important role in digestion and absorption by secreting pancreatic juice, insulin, glucagon, somatostatin, and other hormones. It was reported that pancreatogenic diabetes (type 3c in the American Diabetes Association classification) is frequent after pancreatectomy, so the influence of pancreatectomy on pancreatic function has received increasing attention.
Central pancreatectomy is selected to achieve curative resection while preserving pancreatic function in selected patients with low-grade tumors, such as IPMN, NET, MCN, and SPN [6,7]. If more than 25% of the pancreas is resected during distal pancreatectomy, exocrine function decreases postoperatively [8], while the incidence rate of new-onset diabetes increases with resection of more than 44% of the pancreas [9]. Because the resected parenchymal volume is smaller with central pancreatectomy than distal pancreatectomy, postoperative pancreatic function is expected to be better after central pancreatectomy. The incidence rate of new-onset diabetes ranges from 9% to 19.2% after distal pancreatectomy [10,11]. It was reported that the incidence rate of new-onset diabetes/impaired glucose tolerance was 0% after central pancreatectomy versus 16% after spleen-preserving distal pancreatectomy [12], while another study found rates of 14% after central pancreatectomy and 46% after distal pancreatectomy [13]. However, none of these studies evaluated changes in the condition of patients over time after surgery based on variables such as body weight or serum albumin. In the present study, body weight, serum albumin, and HbA1c were monitored up to 60 months after pancreatectomy to assess both short-term and long-term changes. We showed that new-onset diabetes was significantly less frequent after central pancreatectomy than after distal pancreatectomy (P = 0.0024), and HbA1c did not deteriorate in the short term or long term after central pancreatectomy. In contrast, HbA1c was significantly elevated at 6 months after distal pancreatectomy and was significantly higher again at 36 months and 60 months compared with 6 months. Thus, for distal pancreatectomy, postoperative values indicated both short-term and long-term deterioration of HbA1c. In patients with type 2 diabetes mellitus, long-term treatment with insulin and metformin leads to improvement of glycated hemoglobin levels [14]. In our patients, HbA1c was significantly elevated at 6 months after distal pancreatectomy, probably due to loss of parenchymal volume, but subsequent progressive elevation of HbA1c despite treatment with insulin and oral hypoglycemic agents seems be a characteristic of pancreatogenic diabetes. Unlike the liver, the pancreatic parenchyma does not regenerate after pancreatectomy, and the decrease of parenchymal volume may influence postoperative HbA1c values. It was reported that the blood glucose level increased after 50% of the β-cells in the pancreas were lost [15]. Therefore, the blood glucose level will increase if sufficient β-cell mass is removed during pancreatic resection. We think not only insulin therapy but also oral hypoglycemic therapy are needed after pancreatectomy, because of the loss of the pancreatic volume and worse of the insulin resistance.
In the distal pancreatectomy group, albumin returned to a similar level to that before surgery at 60 months postoperatively and body weight was decreased (although not significantly). In contrast, the albumin level was significantly improved over the long term in the central pancreatectomy group and body weight was increased (although not significantly). These findings suggested that central pancreatectomy is more effective than distal pancreatectomy for preserving both short-term and long-term pancreatic function and nutritional status.
After distal pancreatectomy, it was reported that the incidence rate of the postoperative pancreatic fistula Grade B + C was about 23% [16]. After central pancreatectomy, the incidence rate of the postoperative pancreatic fistula Grade B + C was 11.5-30% [17,18]. In this study, the incident rate of the postoperative pancreatic fistula grade B + C was no significant difference. We think it is equal safety-levels between central pancreatectomy and distal pancreatectomy.
This study had the following limitations: it was a retrospective investigation at a single institution and replacement of pancreatic digestive enzymes was performed actively. There was no significant difference in the incidence of fatty liver between the two groups in the present study, probably because replacement of pancreatic digestive enzymes was initiated before development of fatty liver in at-risk patients. It was reported that the incidence rate of fatty liver after pancreaticoduodenectomy as 37% and fatty liver after pancreaticoduodenectomy was significantly associated with the resection line which meant remnant pancreatic volume [19]. And fatty liver has a potential risk factor for cirrhosis and hepatocellular carcinoma [20], so not providing prevention/treatment for fatty liver is unacceptable. Accordingly, to confirm the present findings, a multicenter prospective study should be performed using an objective method.
Central pancreatectomy was effective for preserving short-term and long-term postoperative pancreatic function and nutritional status. After central pancreatectomy, no patient developed fatty liver, required replacement of pancreatic digestive enzymes, or developed diabetes, so central pancreatectomy was superior for preserving pancreatic function compared with distal pancreatectomy. And there was no significant difference of the incidence rate of the postoperative pancreatic fistula. In addition to the curability of the primary disease, these findings should be taken into consideration when selecting the method of pancreatectomy.
None.
No grant support was provided for this study.
All authors revised the manuscript for important intellectual content, approved the final version, and agree to be accountable for all aspects of the work.
Nothing.