The Anterior Cruciate Ligament (ACL) is frequently assessed using the Lachman test. This test involves linear translation of the tibia on the femur. Meta-analysis and systematic reviews report a wide range of diagnostic accuracy. The purpose of this case report was to establish a proof of concept using a novel device, the Mobil-Aider™, to accurately quantify linear translation of the tibia on the femur.
Radiographs were taken at baseline and in maximal anterior translation of the tibia in a healthy knee. The Mobil-Aider device was strapped on the knee and used to quantify the translation.
The digital reading of the Mobil-Aider was 7.10 mm while the difference between the radiographs measured 6.96 mm.
This report demonstrates a proof of concept. It used a known measure from a radiograph to demonstrate the ability of the Mobil-Aider device to accurately measure the linear translation of the tibia on the femur, i.e. a Lachman test.
Despite being a single case report, this evidence can begin to establish a body of knowledge to quantify joint mobility and assess injury.
Joint translation, ACL laxity, Knee sprain
The Anterior Cruciate Ligament (ACL) is assessed via a widely accepted clinical test: Lachman test [1-5]. This test involves linear translation of the tibia on the femur, i.e. anterior movement of the tibia on the femur in the sagittal plane. Meta-analysis and systematic reviews reported a wide range of diagnostic accuracy [6,7]. Individual modifications of the test and examiner experience have been reported to influence test accuracy [8,9]. Nonetheless, the Lachman test is considered the gold standard [5]. It is performed in the supine position with the knee in 20-30° of flexion.
One study found the misdiagnosis of acute ACL injuries by emergency room physicians to be 74% [10]. There are many reasons for this statistic. When there is a mismatch between the girth of the patient's leg and the size of the clinician's hand, it can be very challenging to perform a Lachman test. If the patient's knee is in too much flexion or if the hamstring musculature is not relaxed, false negative results may be obtained. Finally, asymmetry greater than 3-5 mm in side-to-side laxity or a soft endpoint is considered abnormal [5]. Even for the most experienced clinician, a criterion of 3 mm is extremely challenging to quantify by "feel."
Magnetic resonance imaging (MRI) is often used for ACL injury diagnosis [11]. The ability of MRI to identify partial ACL tears has been called into question [12]. Furthermore, MRI is a static image. Over the past three decades, there have been a few instruments reported to be able to assess dynamic ACL laxity. However, all of the devices have been met with challenges. The Hall Effect Strain Transducer (HEST) was implantable [13]. The Rottometer was a computer-assisted goniometer used to measure rotation of the tibial axis [14,15]. The Vermont knee laxity device was very bulky and required a significant amount of time to utilize [12]. The Lars rotational laxiometer was dependent on too many variables. The Kinematic Rapid Assessment (KiRA) requires Bluetooth technology to measure acceleration of the tibia on the femur [16]. The Vernier Dial Test Indicator has not been validated [17]. The Telos, GNRB, and KT1000/2000 require considerable set up time and do not involve direct clinician contact with the patient [18-20]. The Telos is used in conjunction with radiographs [13,18,21,22]. The GNRB sells for $13,800 and the KT1000/2000 is no longer being produced (only available through the re-sale market since 2012). Several studies have reported substantial variability in the measures using the KT1000/2000, as high as a 28% false-negative rate [17,23s-25].
The Mobil-Aider™ (Therapeutic Articulations, LLC, Spring City, PA USA; info@iortho.xyz) is an orthopedic tool which has evolved from prototype to pre-market level from August 2016 to 2019 (Figure 1). Recent bench testing reported excellent Pearson correlation coefficients (> 0.989), high reliability (Cronbach Alpha > 0.992), and numerous other statistics to support the concurrent validity and reliability of the device (Gulick, 2019 AAOS Conference). The purpose of this case report was to establish a proof of concept using a novel device, the Mobil-Aider™, to accurately quantify linear translation of the tibia on the femur in vivo (Figure 2; "x" indicates the stabilization of the distal femur; "↑" indicates the direction of linear translation).
 Figure 1: Mobil-Aider Device. View Figure 1
Figure 1: Mobil-Aider Device. View Figure 1
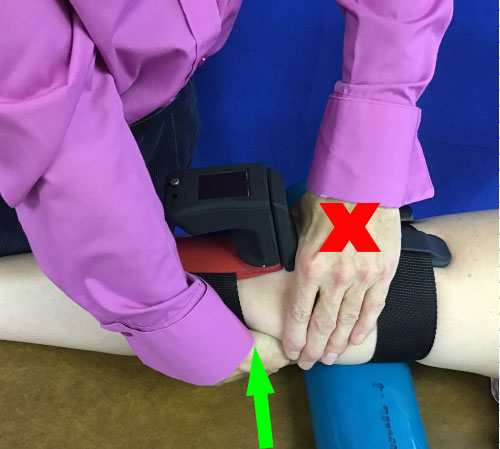 Figure 2: Mobil-Aider on the Knee. View Figure 2
Figure 2: Mobil-Aider on the Knee. View Figure 2
The Mobil-Aider™ is an orthopedic tool used to measure linear translation of a joint. The ability to move in one-plane assures the technique is performed correctly. The device weighs less than 0.4 kg (13 ounces) and includes straps on the femur and tibia to stabilize the device during use. The axis of the Mobil-Aider device was aligned with the joint line of the knee. The knee joint line was defined as the space between the most distal femoral condyles and the most proximal aspect of the tibial plateau. The femur and tibia straps were secured. A lateral knee radiograph was taken in side-lying at rest. A Lachman test was performed and another lateral knee radiograph was taken at the end-range of the Lachman test. The digital reading from the Mobil-Aider was recorded at end-range anterior translation by a third party (examiner was blinded until after the radiograph calculation was performed). Lines were drawn on both radiographs (baseline Figure 3; end-range translation Figure 4) along the most anterior aspect of the tibial plateau and the most anterior aspect of the femoral condyle. The distance between these two lines was measured in the sagittal plane.
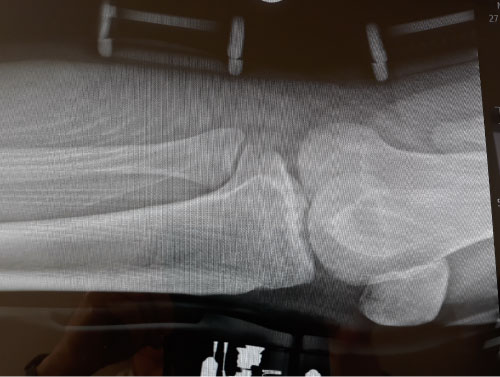 Figure 3: Baseline Radiograph. View Figure 3
Figure 3: Baseline Radiograph. View Figure 3
 Figure 4: Lachman Radiograph. View Figure 4
Figure 4: Lachman Radiograph. View Figure 4
The anterior translation measured from the radiographs was 6.96 mm. The reading on the Mobil-Aider was 7.10 mm. This represents just a 2% difference between the radiographic measures and that of the Mobil-Aider device.
The ability to measure the tibial translation of the knee with a portable, hand-held device could be very valuable in determining the presence and/or the magnitude of an injury to the ACL. Despite the lack of normative values for tibial anterior translation, the ease of comparing the involved to uninvolved knee measure makes this technique clinically relevant. In other words, in the case of a knee injury, a measure of the uninvolved knee would serve as the "normal" for any given individual. A significant advantage of this device, as compared to others, is the technique used does not deviate from that of a "classic" Lachman test. Thus, the clinician has direct contact with the patient and is able to appreciate the joint "end-feel" while obtaining a quantitative value of linear translation.
Although this report only included one radiographic series, it does imply the ability of the device to accurately measure the tibial translation in the sagittal plane. This may be one of several signs and symptoms used to assess ACL injury. As in any study, addressing the limitations using 2D imaging and a single sample can be mitigated with advanced imaging and additional subjects. This research is in process. Likewise, concern over soft tissue compression influencing the measurement of the linear translation deserves mention. However, when used in a clinical setting where a Lachman test can be compared bilaterally, the soft tissue issue will be negated.
Furthermore, the Mobil-Aider can be used to measure the translation of the shoulder, elbow, wrist, and ankle. Measuring linear translational of these joints can also yield valuable information about the laxity/stability of these joints. The ability to quantify joint translation can begin to develop a body of knowledge on which to base decisions regarding evidence-based practice.
The proof of concept/development of this device was funded, in part, by grants from the National Science Foundation (Phase 1), Ben Franklin FabNet, Innovation Partnership, and Singh Nanocenter.
The author is awaiting patent approval of this device. It is FDA approved as a class 1 exempt measurement device.