Acute lymphoblastic leukemia (ALL) is an unusual diagnosis in the adult population. These cases are rarely reported in combination with ischemic stroke and endocarditis. We present a case of an elderly female patient presenting with acute hyperleukocytosis and new diagnosis of ALL, border-zone ischemic stroke and nonbacterial thrombotic endocarditis.
81-year-old female presented to an outside emergency room with subacute onset of left facial weakness and right hemiparesis for 12 hours. A non-contrast CT scan of the head and CTA head and neck showed no acute findings. Laboratory results were remarkable for a leukocyte count of 245.75 k/uL. She was diagnosed with acute leukemia and transferred to a comprehensive stroke center where she was treated with leukapheresis and hydroxyurea. The patients focal deficits somewhat improved, but in the following days, became confused and aphasic. Brain MRI showed bilateral acute border-zone ischemic infarcts (Figure 1). Echocardiogram was also ordered as part of the routine stroke work up. It detected a mobile mass on the ventricular side of the mitral valve leaflet concerning for nonbacterial thrombotic endocarditis. The patient was a febrile and blood cultures were negative on four repeated occasions, seven days apart. The family chose not to pursue any further invasive work up given her aforementioned wishes, and she was eventually discharged to home hospice with comfort care with palliative care team consultation.
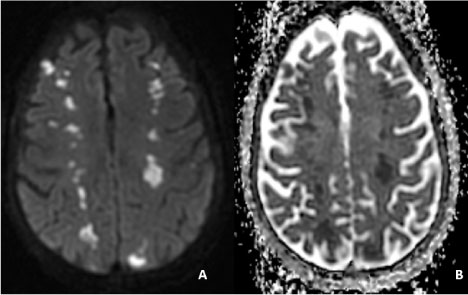 Figure 1: Brain MRI without contrast.
Figure 1: Brain MRI without contrast.
These images demonstrate a hyperintensity in the diffusion weighted image (A) and hypointensity in the apparent diffusion coefficient image (B) in bilateral hemispheres consistent with acute ischemic infarction. These lesions are located in the frontal lobes at the border zones of the MCA-ACA territories and parieto-occipital region at the limits of the MCA-PCA territories. View Figure 1
To the best of our knowledge, this case illustrates the first report of an adult patient diagnosed with ALL in combination with an acute border-zone ischemic stroke and nonbacterial thrombotic endocarditis. It is important to recognize that acute leukemia scan have an effect on cerebral perfusion and may present with hyper-viscosity leading to ischemic cerebrovascular and cardiac events.
Acute lymphoblastic leukemia, Hyperleukocytosis, Hyperviscosity, Border-zone stroke, Watershed stroke, Endocarditis
ALL: Acute Lymphoblastic Leukemia; AML: Acute Myeloid Leukemia; ACA: Anterior Cerebral Artery; MCA: Middle Cerebral Artery; PCA: Posterior Cerebral Artery; IE: Infective Endocarditis; NBTE: Nonbacterial Thrombotic Endocarditis
Acute lymphoblastic leukemia (ALL) is an unusual diagnosis in the adult population. They represent 20% of the cases in the adult population. Moreover, acute leukemia in the elderly is rarely reported in combination with ischemic stroke. We present a case of a female patient presenting with acute hyperleukocytosis and new diagnosis of ALL, border-zone ischemic infarcts, and nonbacterial thrombotic endocarditis.
An 81-year-old female with past medical history significant for stable congestive heart failure, atrial fibrillation on apixaban, and coronary artery disease presented to an outside hospital with 12-hour history of right-sided hemiparesis and contralateral facial droop. On presentation, speech was without significant dysfluency. Physical exam revealed a heart rate of 105 but otherwise the patient was hemodynamically stable in no acute distress. On neurologic exam, she exhibited left sided facial weakness and right sided diminished sensory and motor strength in upper and lower extremities. There were no acute findings on non-contrast CT head or CT angiogram of head and neck. ECG revealed atrial fibrillation with mild rapid ventricular rate. White blood cell count was remarkably elevated at 245.75 k/uL with immature granulocytes and blasts. Troponin was also elevated. She was suspected to have hyperviscosity-induced neurologic symptoms from acute leukemia and transferred for a higher level of care.
She was given hydroxyurea and underwent leukapheresison hospital day one. Her post-procedure leukocyte count reduced to 97.8 k/uL. She was treated with a four-day course of dexamethasone and continued treatment with hydroxyurea with a marked improvement of leukocyte count. Flow cytometry revealed results consistent with B-cell acute lymphoblastic leukemia.
Clinically the patient became increasingly confused and aphasic with a waxing and waning course on neurologic exam. She inconsistently followed simple commands. Physical exam also showed Janeway lesions in finger tips and toes. A repeat CT head performed 24 hours post initial imaging revealed interval development of multiple bilateral cortical and subcortical hypodense areas in parietal and occipital lobes consistent with ischemic infarction. MRI confirmed bilateral watershed infarcts in the ACA/MCA and MCA/PCA territories (Figure 1). An echocardiogram with contrast was obtained revealing ejection fraction of 55-59%, with left and right atrial enlargement, and a non-enhancing globular mobile mass measuring 1.7 cm × 1.7 cm attached in the ventricular side of the posterior mitral leaflet. The cardiologist interpretation of the mass was that of marantic endocarditis given its contrast appearance, location and clinical scenario. Four sets of blood cultures were negative, including a repeat after seven days, and patient did not develop a fever during the hospital course. Four days later, the echocardiogram was repeated and showed no changes in size of the mass. Further invasive evaluation, including trans-esophageal echocardiography was deferred by the patient's family.
For the remaining hospital course, her neurologic exam did not improve significantly. Given her co-morbid conditions and guarded prognosis, her family decided not to pursue future chemotherapy. She was discharged to hospice care with palliative care consultation.
Acute leukemias are characterized by a rapid increase in white blood cells. These can be divided into lymphoblastic or myeloblastic. Acute lymphoblastic leukemia is uncommon in the elderly population and represents approximately 20% of patients above the age of 55 [1]. These patients do not respond as well as the younger population to chemotherapy, and an inverse correlation between age and survival has been reported [1]. Most cases of acute stroke presentation are reported in acute myeloid leukemia (AML), which remains the most common type of acute leukemia in adults. This case highlights an unusual case of ALL in an adult patient, in combination with uncommon findings of border-zone cerebral ischemic infarcts and mitral valve mass.
The prevalence of ischemic strokes in adults with ALL is unknown. In the pediatric population, some studies have reported a 0.47% prevalence with most being related to sinovenous thrombosis [2]. However, epidemiologic studies have reported a 0.59% prevalence of cerebrovascular accidents in a studied population of 10,974 patients diagnosed with AML [3]. The pathophysiology of ischemic events in acute leukemias may be related to the hyperleukocytosis and leukostasis. Hyperleukocytosis is defined as a total leukocyte count of > 100 k/uL [4]. If hyperleukocytosis presents with associated decreased tissue perfusion, it is termed leukostasis. The incidence of hyperleukocytosis is 10 to 30% in newly diagnosed ALL [5]. The increase in lymphoblasts can affect blood viscosity, and subsequently blood flow, leading to hypoxemia [6]. Therefore, in the appropriate setting, patients with ALL can present with signs and symptoms of ischemia leading to acute stroke or myocardial infarction [7].
The ischemic lesions seen on brain MRI follow the pattern of border-zone infarcts, also referred to as watershed territory. Pathophysiologically, the ischemic changes seen are due to a decrease in global perfusion, particularly affecting vulnerable tissue in distal regions where two vascular territories meet. The hemodynamic alterations can be related to an area of stenosis in the setting of hypotension, or rarely a compromise in cerebral blood flow due to a hemodynamically hyperviscous state. This type of stroke pattern has been reported in certain cases of leukemias, including hypereosinophilic subtype [8].
Another cardiac abnormality which can be seen in leukemias is endocarditis. There have been various reports of infective endocarditis (IE) in patients with ALL and AML. Nevertheless, cases of nonbacterial thrombotic endocarditis (NBTE), or marantic endocarditis, are few by report in AML, and extremely rare in ALL [9]. They are associated with several other hematologic conditions, such as hypercoagulable states which can be seen with various forms of malignancy [10]. Patients with cancer have elevated cytokines, which causes local tissue damage and can promote vegetation formation [11]. Our patient had an echocardiogram with contrast which showed a non-enhancing mobile mass attached to the ventricular side of the posterior mitral leaflet, concerning for possible NBTE. Blood cultures were negative on four sets, and after repeat one week later. Additionally, the patient did not have a fever during the entirety of her admission. Infective endocarditis is typically found on the auricular surface of auriculo-ventricular valves [10]. Images of the echocardiogram obtained during admission were also compared to a prior normal echocardiogram completed only five months prior to the present admission. On clinical exam, the patient also had Janeway lesions in the palms and soles, indicative of systemic embolization which is more common in NBTE than IE in the western world [10]. On the contrary, vasculitis phenomena such as Roth spots, glomerulonephritis and splinter hemorrhages are more common in IE [10].
We present a rare case of an elder adult with acute hyperleukocytosis diagnosed with acute lymphoblastic leukemia presenting as an acute ischemic stroke. It is important to remember the possible association with ischemic strokes due to the hemodynamic changes and decreased cerebral flow in the setting of hyperviscosity in patients with leukemia. Additionally, cardiac abnormalities can be seen as was in our patient who presented with endocarditis thought to be triggered by a secondary hypercoagulable state. To our knowledge, this is the first case to report the combination of these three entities.
Authors' original file for Figure 1 MRI brain without contrast.
The authors declare that they have no competing interests.
All authors contributed to writing the manuscript. All authors read and approved the final manuscript.