Solitary fibrous tumors (SFTs) typically arise from the pleura and other thoracic locations but can also rarely affect the nasal cavity and paranasal sinuses. This report describes the presentation, workup, and management of a patient with a sinonasal SFT with expansion into the intracranial fossa.
Case report and literature review.
We report on a 78-year-old male who presented with approximately 1 year of worsening nasal obstruction, difficulty sleeping, and loss of olfaction and gustation. Fiberoptic examination, CT, and MRI all demonstrated a mass of the right nasal cavity that was consistent with a SFT on pathologic and immunohistologic evaluation. The mass was completely resected through an endoscopic endonasal approach without post-operative complications.
Although SFT are rare tumors that present in the sinonasal cavities, awareness of this pathologic entity and its histopathologic features enhances the workup for diagnosis and treatment of nasal masses.
Solitary fibrous tumor, Sinonasal cavity, Benign tumor
Solitary fibrous tumors (SFTs) are rare masses of mesenchymal origin that were first described by Klemperer and Rabin in 1931 and initially thought to be of mesothelial origin. SFTs are typically found originating from the pleura and other intrathoracic or peritoneal locations [1]. However, because of their mesenchymal origin, they have been reported originating from many different anatomic locations. SFTs are found in the head and neck in up to 5-27% of reported cases, most commonly the oral cavity and orbit [2]. Additional locations reported include the external auditory canal, lacrimal sac, epiglottis, larynx, thyroid, sublingual gland, parotid gland, tongue, gingiva, orbit, parapharyngeal space, nasopharynx, hypoglossal nerve, scalp, and infratemporal fossa [3].
SFTs of the nasal cavity and paranasal sinuses are extremely rare, and to our knowledge, there is limited literature available concerning the presentation, diagnosis, and treatment of sinonasal SFTs. Here, we describe the case of a 78-year-old male with a SFT of the right nasal cavity extending into the cranial vault, paranasal sinuses, and nasopharynx.
A 78-year-old male was referred for otolaryngologic evaluation with approximately 1 year of progressively worsening right nasal obstruction. The patient presented with difficulty sleeping at night due to trouble breathing through his nose and gradual loss of olfaction and gustation for about 4 months. On nasal endoscopy, a well-mucosalized lesion from the cribriform plate to the inferior nasal cavity was visualized (Figure 1E). The nasal septum demonstrated a broad deflection to the left, resulting from the mass effect produced by the sinonasal mass.
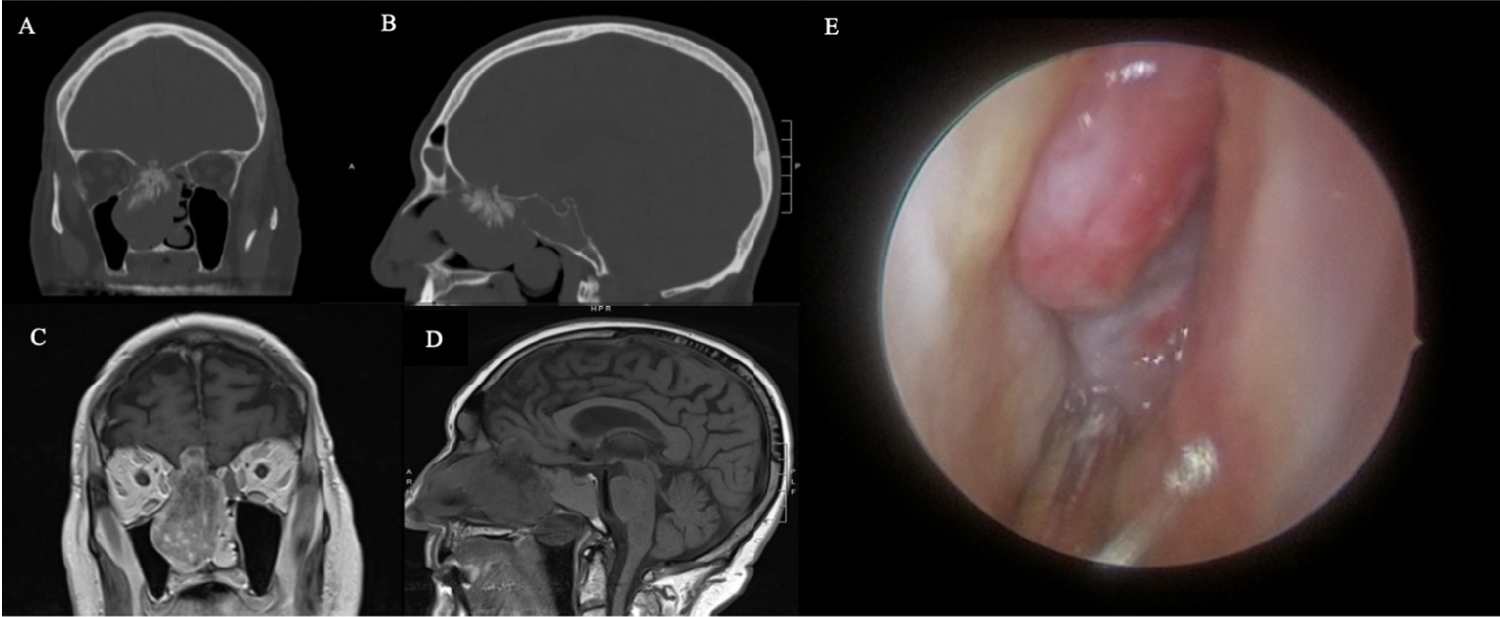 Figure 1: Pre-operative Assessments. On non-contrast CT, the coronal (A) and sagittal (B) views of the right sinonasal solitary fibrous tumor demonstrate a calcified root along the right cribiform plate and opacification of the adjacent frontal, ethmoid, and sphenoid sinuses. T1-weight magnetic resonance imaging shows the right heterogeneous sinonasal mass with intracranial extension into the right frontal lobe in coronal (C) and sagittal (D) views. On nasal endoscopy, the right sinonasal solitary fibrous tumor is well-mucosalized and originates from the anterior skull base (E).
View Figure 1
Figure 1: Pre-operative Assessments. On non-contrast CT, the coronal (A) and sagittal (B) views of the right sinonasal solitary fibrous tumor demonstrate a calcified root along the right cribiform plate and opacification of the adjacent frontal, ethmoid, and sphenoid sinuses. T1-weight magnetic resonance imaging shows the right heterogeneous sinonasal mass with intracranial extension into the right frontal lobe in coronal (C) and sagittal (D) views. On nasal endoscopy, the right sinonasal solitary fibrous tumor is well-mucosalized and originates from the anterior skull base (E).
View Figure 1
A noncontrast computed tomography (CT) scan showed a partially calcified mass measuring 5.3 cm by 3.5 cm by 5.7 cm in the region of the right cribriform plate, filling the right nasal passage and extending into the inferior aspect of the right frontal lobe (Figure 1A and Figure 1B). Complete opacification of the right frontal sinus, right ethmoid air cells, and right sphenoid sinus were noted along with deviation of the nasal septum to the left. Magnetic resonance imaging (MRI) with and without intravenous contrast was also obtained, which showed a heterogeneously enhancing calcified mass consistent with that seen on CT (Figure 1C and Figure 1D). MRI showed expansion of the mass into the extra-axial sub-frontal space abutting the gyrus rectus and medial orbitofrontal gyrus. Expansion into the right maxillary sinus, right sphenoid sinus, ethmoid cells, and right medial orbital extraconal space without intra-orbital invasion was also seen on MRI.
An endoscopic biopsy of the right nasal mass was completed under local anesthesia. Two separate pathology laboratories evaluated the specimen and determined that the cytology and immunohistochemistry were most consistent with SFT. Histologic evaluation of the mass showed a pattern-less submucosal spindle cell population comprised of uniform bland cells with vesicular-nuclear chromatin and scant-to-moderate cytoplasm. The spindle cells were interspersed within a background of stromal sclerosis with numerous vessels observed. Although the tumor showed an infiltrative growth pattern, mitotic activity was low, and cellularity was not significantly increased (Figure 2). Thus, the tumor was determined to be non-malignant. The mass was positive for CD99 and STAT6. Interestingly, the sample was negative for CD34, which is a rare finding for SFTs of any anatomic location. The sample was also negative for SMA, beta-catenin, CD163, Factor XIIIa, EMA, pan-cytokeratin, S100, SOX10, and pan-melanin.
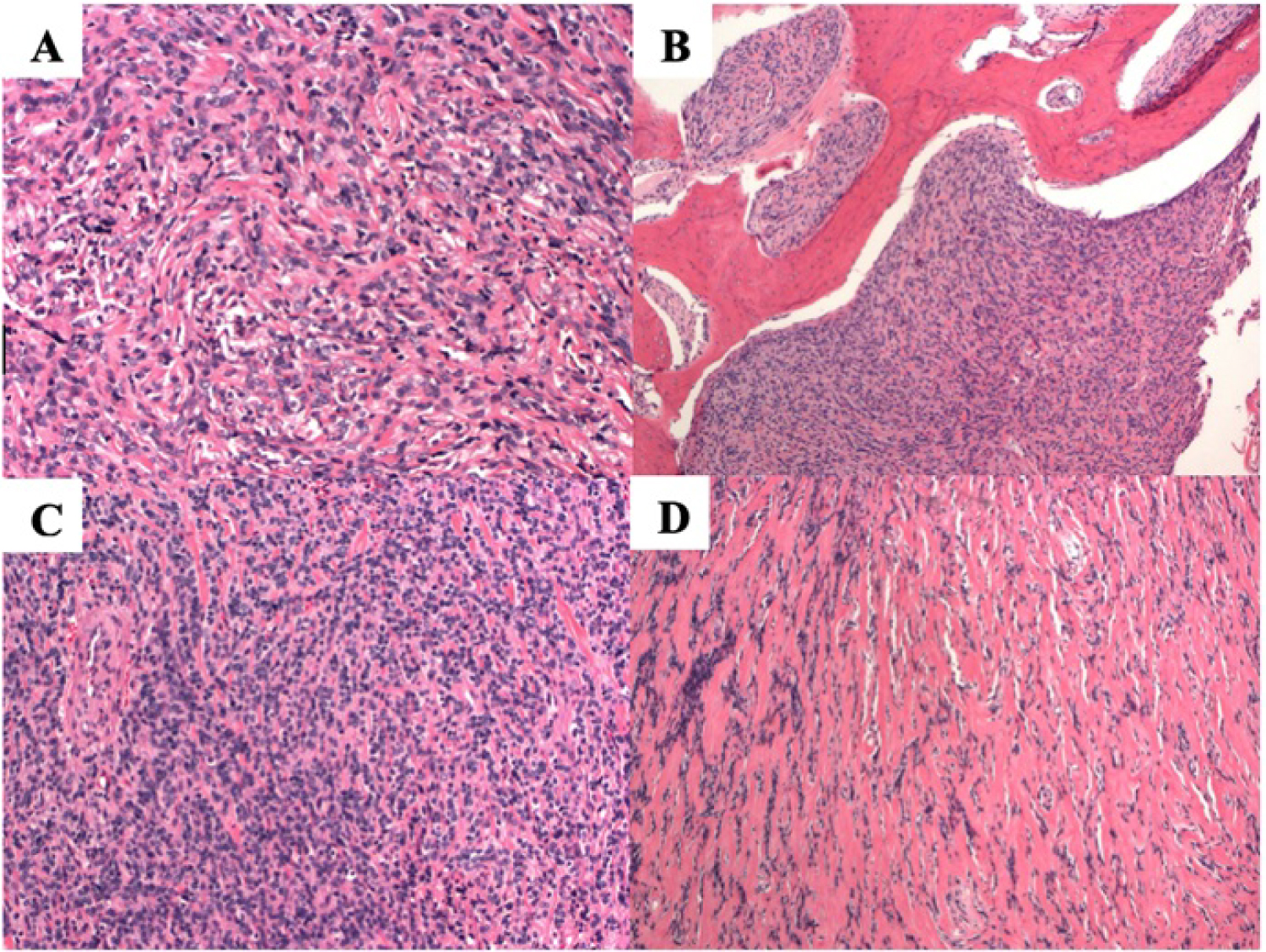 Figure 2: Histologic examination from intra-operative sample prepared with hematoxylin and eosin stain. This tumor demonstrates a "patternless" pattern of bland uniform spindle cells, separated by mature pink collagen (A). There is pushing erosion into bone, filling spaces between bone trabeculae (B). Variable cellularity can be seen with this tumor ranging from dense cellularity with little collagen (C) to sparse cellularity and abundant hyalinized pink collagen (D).
View Figure 2
Figure 2: Histologic examination from intra-operative sample prepared with hematoxylin and eosin stain. This tumor demonstrates a "patternless" pattern of bland uniform spindle cells, separated by mature pink collagen (A). There is pushing erosion into bone, filling spaces between bone trabeculae (B). Variable cellularity can be seen with this tumor ranging from dense cellularity with little collagen (C) to sparse cellularity and abundant hyalinized pink collagen (D).
View Figure 2
The patient underwent surgical resection through an endoscopic endonasal approach with navigation assistance. Drilling of the bony aspects of the sinonasal tumor was required, but the sinonasal and intracranial components of the mass were completely excised with a four-hand technique. Intraoperative frozen tissue specimen of deeper aspects of the right nasal mass confirmed the diagnosis of SFT. The anterior skull base defect was repaired with a non-synthetic dura substitute (Durepair Regeneration Matrix, Medtronic, Minneapolis, MN) and a free mucosal graft taken from the ipsilateral nasal floor. The patient's post-operative course was uncomplicated with immediate improvements in his pre-operative nasal obstruction and sleep quality. A post-operative MRI showed the complete resection of the right sinonasal tumor (Figure 3A and Figure 3B), and the patient remains clinically disease-free 8 months following surgery (Figure 3C).
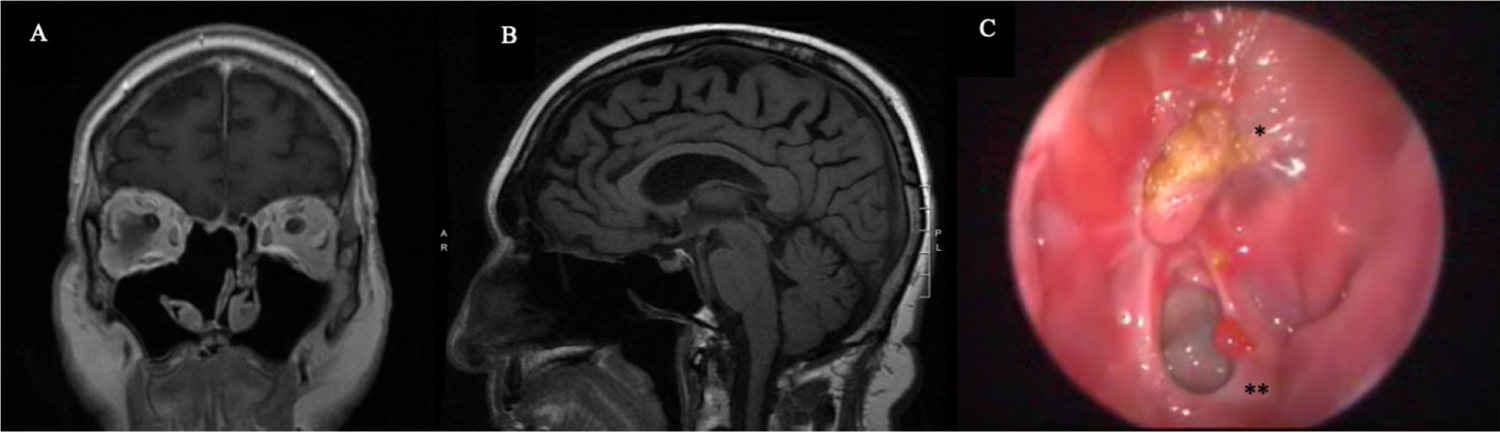 Figure 3: Post-operative Assessments. T1-weight magnetic resonance imaging shows adequate resection of the right sinonasal mass at 3 months following surgery in both coronal (A) and sagittal views (B). Nasal endoscopic exam at 8 months following surgery (C) reveals a well-healed right anterior skull base (*) and a patent right sphenoid ostium (**).
View Figure 3
Figure 3: Post-operative Assessments. T1-weight magnetic resonance imaging shows adequate resection of the right sinonasal mass at 3 months following surgery in both coronal (A) and sagittal views (B). Nasal endoscopic exam at 8 months following surgery (C) reveals a well-healed right anterior skull base (*) and a patent right sphenoid ostium (**).
View Figure 3
While SFTs are mesenchymal tumors that are most commonly found on the pleura and in the thoracic cavity, such tumors may rarely occur in head and neck region and in the sinonasal compartments. Most documented cases of SFTs in the sinonasal cavities are noted to be asymptomatic. Among symptomatic patients, symptoms include nasal obstruction, rhinorrhea, congestion, ptosis, and exophthalmos. The tumor is generally painless and slow-growing and has an extremely low risk of malignancy.
On CT imaging, SFT has been described as having isoattenuation with the surrounding musculature and soft tissue. This attenuation increases with the administration of intravenous contrast. Rare findings of calcification, as seen in our reported patient, and intratumoral low attenuation areas have also been identified. The low attenuation areas are most likely indicative of myxoid or cystic degeneration. On T1-weighted MRI, SFTs are isointense to the surrounding tissue. However, T2-weighted MRI images are more heterogeneous depending on the character of the tumor such as the amount of collagen and fibroblasts or the presence of degeneration. The intensity of the signal on both T1- and T2-weighted MRI increases with the administration of intravenous contrast. The reason for increased signal on CT and MRI with contrast is due to the highly vascular nature of the SFT [4,5].
On histology, SFTs are composed of proliferations of spindle cells dispersed within a collagen-rich stroma (Figure 2A). The cellularity is usually variable with areas of hypercellularity as well as hypocellular areas (Figure 2C and Figure 2D) consisting of hyalinized stroma with rare spindle cells. The tumors are highly vascular with many small to medium-sized thin-walled vessels as well as larger, thick-walled vessels. The neoplastic cells do not usually display signs of atypia [2].
The most sensitive and specific means for diagnosing an SFT is through immunohistochemical studies. CD34 and Bcl-2 are the most sensitive first-line markers for the diagnosis of SFT. CD34 is positive in 95-100% and bcl-2 is positive in 50-100% of documented cases of SFTs depending on the location in the body [6]. Thus, a tumor negative for CD34 and bcl-2 is unlikely to be SFT. It should be noted, however, that the rate of positive CD34 is decreased to about 83% in cases of malignant SFT. In our reported patient, however, the sinonasal mass was negative for CD34; this finding constitutes an immunohistochemically rare presentation of SFT. Our patient nonetheless demonstrated high expression of CD99 and STAT6, which are additional markers that are useful for diagnosis of SFT. CD99 is positive in about 87% of SFTs [7]. Intense staining of STAT6, which represents the NAB2-STAT fusion gene, is seen in more than 90% of cases of SFT, while less than 10% of other spindle cell tumors stain positive for STAT6 and these other tumors usually stain fair less strongly than SFTs [8,9].
The differential diagnosis of a SFT presenting as a sinonasal mass is fairly broad and should include other benign and malignant tumors. Sinonasal glomangiopericytoma is similar to SFT in that it is composed of plump spindle cells interspersed with numerous branching vessels [10]. However, these tumors do not display the dense, hyalinized collagen that is typical of SFT. SFT and glomangiopericytoma can also be differentiated based on immunohistochemical staining. Glomangiopericytomas are smooth muscle tumors and stain positive for muscle specific actins. These tumors also typically do not stain for STAT6 and CD34, which are characteristic of SFTs [11-13].
Another tumor that can appear similar to SFT is a nasopharyngeal angiofibroma. These tumors are made up of a collagen-rich stroma with many branching blood vessels, similar to the appearance of an SFT. However, these angiofibromas will usually stain for androgen receptors and do not exhibit the STAT6 and CD34 markers characteristic of SFTs [14].
Angioleiomyoma should also be included on the differential with SFT. These tumors are composed of intersecting fascicles of spindle cells with numerous blood vessels. As with the last two tumors discussed, angioleiomyomas do not stain for STAT6 or CD34. Instead, they can be stained for SMA, MSA, h-caldesmon, and desmin [15,16].
Nerve sheath tumors such as schwannoma and neurofibroma can also rarely be found in the nasal cavity and paranasal sinuses and share particular characteristics with SFTs. Nerve sheath tumors stain positive for neural markers such as S-100 and SOX10, both of which were negative in the patient described in this report. Unlike the tumors discussed above, some nerve sheath tumors may also stain positive for CD34, but can be differentiated from SFT because they will always be STAT6 negative [17].
SFTs can also be mistaken for some sarcomas because sarcomas may be composed of spindle cells. In particular, synovial sarcoma, mesenchymal chondrosarcoma, and biphenotypic sinonasal sarcomas can all arise in the sinonasal tract and mimic SFTs. These sarcomas will all be negative for STAT6 on immunohistochemical staining. Some biphenotypic sinonasal sarcomas can be positive for CD34, but a majority will be negative and both synovial sarcoma and mesenchymal chondrosarcoma will always be negative for CD34 [2].
Complete surgical resection is recognized as the best definitive treatment for SFTs. Tumor diameter larger than 10 cm, the presence of a malignant component to the histologic findings, and microscopically positive surgical margins are all associated with local recurrence after surgical intervention [18]. In prior case series, all head and neck SFTs were small (< 10 cm) and local recurrence was primarily due to the lack of complete resection with clear margins. This is likely because of the challenging anatomy of the head and neck and proximity to other local structures [1]. Adjuvant therapy is typically not necessary, especially since recurrence is slow and usually non-malignant. However, use of radiation or chemotherapy has been used to manage locally aggressive or recurrent disease in rare cases [19,20].
SFTs, in general, have a relatively good prognosis with metastasis or recurrence being extremely rare. The World Health Organization (WHO) characterizes SFTs as malignant if they display hypercellularity, increased mitoses (> 4 mitoses per 10 high power fields), cytological atypia, tumor necrosis, and/or infiltrative margins [21]. SFTs exhibiting these characteristics are more likely to recur and are associated with decreased survival. Based on these criteria, the tumor isolated from the patient described above was non-malignant. There have been models developed to help assess the risk of SFTs based on age, tumor size, and number of mitotic figures [8]. Among all SFTs, regardless of anatomic location, the rate of malignant tumor has been reported at 10-20%. However, malignancy is rare in the sinonasal tract. There have been no previously reported cases of metastasis or death in patients with SFT of the sinonoasal tract [2]. Although a vast majority of the previously reported cases of sinonasal SFTs behave in a benign and predictable fashion, some will locally recur. Prognostic details for sinonasal SFTs are lacking due to the limited number of documented cases with adequate follow up.
The diagnosis of SFT of the sinonasal cavity relies on the aggregation of multiple clinical data points. Although immunohistochemical markers are the most accurate means of diagnosis, they are not perfect. In this case, the patient's tumor was negative for CD34 which is very rare in the case of SFT. Thus, it was important to take into account other markers, both positive and negative, and the clinical characteristics of the sinonasal mass. Once a diagnosis is established, complete resection of the tumor is the most widely accepted treatment for SFT and adjuvant chemotherapy or radiation is usually not necessary.