The aim of this study was to evaluate the frequency of hyperkalemia in chronic kidney disease patients with eGFR < 60 ml/min/1.73 m2 and under regular nephrologic follow-up.
286 patients were studied. Patients were divided into two groups according to the serum potassium as normokalemic group (n = 184) and hyperkalemic group (n = 97). Demographic and labarotory properties in normokalemic group and in hyperkalemic group were compared.
The prevalance of hyperkalemia was 33.9% in study population. There was no significant difference in serum potassium level between chronic kidney disease stages (p = 0.589).
This study showed a high prevalence of hyperkalemia among patints with chronic kidney disease under regular nephrologic follow-up.
Hyperkalemia, Chronic kidney disease, Normokalemia
Hyperkalemia is an important life-threatening electrolyte disorder and one of the most important complications of Chronic Kidney Disease (CKD). Decreased Potassium (K) excretion due to decreased glomerular filtration rate and reduced tubular secretion due to tubulointerstitial dysfunction lead to abnormal K balance and hyperkalaemia in CKD [1]. In addition, the use of Renin Angiotensin Aldosterone System (RAAS) blockers, which are important in the treatment of CKD, and the presence of Diabetes Mellitus (DM) also cause hyperkalemia by decreasing urinary potassium excretion [2-4]. Metabolic acidosis associated with renal failure leads to potassium shift from intracellular to extracellular and thus hyperkalemia [5]. Most of the daily potassium is excreted by the kidneys while the remaining 5-10% is eliminated by the colon [6]. When kidney failure develops, the amount of K excreted by the urine decreases while the amount of K excreted by the colon increases. However, this is usually not enough to keep serum K level within normal limits [6].
Serum K level is regulated between 3.5-5.0 mEq/L. Regulation of serum K level in this range is important in terms of cardiac conduction, cardiac contraction, smooth muscle tone and neuronal transmission [7,8]. Hyperkalemia is defined as serum K above 5.0 or 5.5 meq/L [9,10]. Severe hyperkalemia causes ventricular arrhythmia and sudden death [10-13].
The aim of this study was to determine the frequency of hyperkalemia in patients with eGFR below 60 ml/min/m2 and not on dialysis that under regular nephrologic follow-up, and to compare the demographic and clinical characteristics of hyperkalemic and normokalemic patients with CKD.
Patients with CKD stages 3-5 (not on dialysis) who were admitted to the nephrology clinic during the three-month period (January-March 2016) were included in the study retrospectively. All patients had at least 3 or more nephrology visits and received information about diet for CKD. Hemodialysis patients and patients with acute deterioration of renal function were excluded from the study. Patients with CKD who were not under regular nephrologic follow-up were also excluded from the study.
Patients' laboratory (serum K, serum creatinine, GFR, hemoglobin, ferritin, parathormone) and demographic characteristics (age, gender, DM, hypertension, concomitant diseases such as heart failure and cause of CKD) were recorded. Patients were evaluated for drug use (RAAS blockers, beta blockers, diuretics, sodium bicarbonate, polystyrene sulphonate) that affected potassium levels.
Patients were divided into two groups according to the results of serum K: Normokalemic group (serum K = 3.5-5.0 mEq/L) and hyperkalemic group (serum K > 5.0 mEq/L). Patients with a serum K level of 6.0 mEq/L or greater were considered severe hyperkalemic. The drugs, demographic and laboratory characteristics of hyperkalemic and normokalemic patients were compared.
Patients were divided into CKD stages: Stage 3A (GFR = 45-59 mL/min/1.73 m2), stage 3B (GFR = 30-44 mL/min/1.73 m2), stage 4 (GFR = 15-29 mL/1.73 m2) and stage 5 (GFR < 15 mL/min/1.73 m2). Serum K levels, hyperkalemia frequency and frequency of using polystyrene sulphonate were compared among the CKD stages.
Statistical analysis was performed using the SPSS 15 program. Chi-square test for intermittent variables and Mann-Whitney U test for continuous variables were used for comparison of normokalemic and hyperkalemic groups. The Kruskal-Wallis test was used to compare serum K levels among CKD stages. A value of p < 0.05 was considered statistically significant. Continuous variables were expressed as mean ± SD, and intermittent variables were expressed as number (%). Spearman correlation analysis and multivariate linear regression analysis were used to evaluate factors that significantly relating with serum K level.
Hyperkalemia was detected in 97 (33.9%) of 286 patients with CKD in this study. Severe hyperkaemia was found in 4.1% of hyperkalemic patients. Only 5 (1.7%) patients had hypokalemia. Demographic and laboratory characteristics of hypokalemic and normokalemic chronic kidney patients were compared in Table 1.
Table 1: Comparison of demographic and laboratory characteristics of Hyperkalemic and Normokalemic groups. View Table 1
81 (28.6%) of the patients were using RAAS blockers. There were no significant differences in the use of diuretics, beta blockers and calcium channel blockers among the hyperkalemic and normokalemic groups. Comparisons of drugs used by hyperkalemic and normokalemic groups are shown in Table 2.
Table 2: Comparison of drugs used in Hyperkalemic and Normokalemic groups. View Table 2
There was a negative but not significant correlation between glomerular filtration rate and serum K value (p = 0.184, r = -0.079). The mean serum K levels of patients according to CKD stages are shown in Table 3 and Figure 1. There was no significant difference in the incidence of hyperkalemia among CKD stages (stage 3A: 28.2%, stage 3B: 36.0%, stage 4: 37%, stage 5: 38.5%, p = 0.625). There was no significant difference in the frequency of polystyrene sulphonate use among CKD stages (stage 3A: 4.2%, stage 3B: 11.5%, stage 4: 11.8%, stage 5: 11.5%, p = 0.345).
Table 3: Mean potassium values according to chronic kidney disease stages*. View Table 3
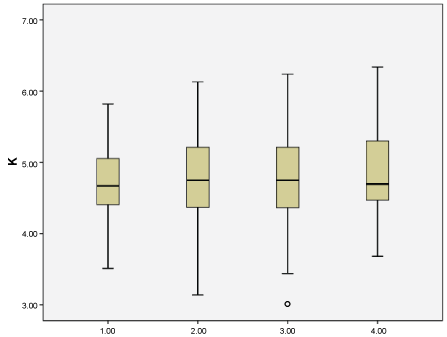 Figure 1: Serum K levels according to chronic kidney disease levels. View Figure 1
Figure 1: Serum K levels according to chronic kidney disease levels. View Figure 1
Correlation analysis was made between serum K level and other continous variables. The effects of serum creatinine (r = 0.122, p = 0.039) and hemoglobine (r = -0.141, p = 0.02) values, which were found to be significant in the correlation analysis, on serum K level were evaluated by multivariate linear regression analysis. Only serum hemoglobine was found to have an independent effect on serum K level (B = -0.141, p = 0.02).
In this retrospective study, it was determined that approximately one-third of the follow-up chronic renal patients were hyperkalemic, but severe hyperkalemia was seen in only 1.3% of the patients. Hyperkalemia in the general population is a rare electrolyte disorder with a frequency of 2.6% to 3.5% [14,15]. The incidence of hyperkalaemia in chronic renal patients varies widely from 7.7% to 73.0% [13,16]. This variability can be explained by the use of different definitions for hyperkalemia and the presence of predisposing factors. There is also a linear relationship between risk of hyperkalemia and low GFR in CKD [15]. In this study, there was a negative correlation between GFR and serum K level but this did not reach significant value. This result may be due to the low number of patients.
A large study investigating the frequency and significance of hyperkalemia in CKD has reported that the frequency of hyperkalemia increases in all patients, although not in the use of RAAS blockade [17]. Gluhovschi, et al. [18] showed that the frequency of hyperkalemia in stage 5 predialysis patients was 37.5% similar to our study. However, in this study serum K levels of 5.5 mEq/L and above were accepted as hyperkalemia. Another study in which serum K levels of patients with a GFR of less than 60 ml/min/1.73 m2 were investigated was found to be 11% with a serum K level above 5.0 mEq/L, which is quite low compared to our study [19]. Factors affecting the frequency of hyperkalaemia were RAAS blocker use, DM presence and low GFR [19]. Serum K levels above 5.0 mEq/L in stage 3 and 4 CKD increased the risk of mortality but did not affect progression to end-stage renal disease [19].
Patients with CKD are at high risk for cardiovascular disease and end-stage renal disease. Drugs that block the RAAS have protective effects on the heart and kidney. However, RAAS blockers increase serum K levels and due to hyperkalemia so these drugs may need to be discontinued, especially in patients with heart failure and CKD, despite the beneficial effects on mortality [20,21]. The use of RAAS blockers in CKD patients is associated with a higher incidence of hyperkalemia and therefore the discontinuation of RAAS blockers is significantly higher than in the general population [21,22]. In our study, the rate of use of RAAS blockers in the hyperkalemic group was lower. This may be related to the inability of the hyperkalemic patients to initiate RAAS blockers or to discontinue RAAS blockers due to hyperkalemia. K/DIGO guidelines recommend the use of RAS blockers in macroalbuminuric patients and in diabetic patients with microalbuminuria. In this study, the percentages of proteinuric patients in the two groups were unknown. So the difference in the proportion of proteinuric patients in the normokalemic and hyperkalemic groups may also lead to difference in the usage of RAS blockers between the two groups. The use of sodium bicarbonate was higher in the hyperkalaemic group. Sodium bicarbonate increases intracellular potassium shift due to improved metabolic acidosis and helps to correct hyperkalemia. Polystyrene sulfonate is a potassium-binding resin and can be used in the treatment of hyperkalemic patients. In this study, the use of polystyrene sulfonate was higher in the hyperkalaemic group. Because of the retrospective nature of the study, no comments were made regarding doses of these drugs and their effectiveness.
There is a U-shaped relationship between serum K level and mortality, both low and high serum K values are associated with mortality [16,23,24]. Significant associations were found between hyperkalemia and increased risk of mortality and arrhythmia in observational studies [17,23,24]. The risk of ventricular fibrillation is increased in patients with a serum K level above 5.0 mEq/L and hospitalized with an acute coronary event [24]. The 1-day-mortality ratio was increased significantly in hospitalized patients with serum K levels above 5.5 mEq/L [17]. In a retrospective study with non-dialyzed CKD patients, when patients were followed for an average of 2.76 years, both hypokalemia and hyperkalemia were found to have a strong, independent, and statistically significant association with mortality, major cardiovascular events, and increased incidence of hospitalization [23].
Limitations of this study are the low number of patients and the lack of interpretation of hyperkalemia and clinical outcomes because of the cross-sectional nature of the study. However, despite the regular follow-up of patients, it is important that the frequency of hyperkalemia is high. This may indicate that patients are inconsistent with the diet.
As a result, hyperkalemia is a common problem in chronic renal patients who do not dialyze and under regular nephrology follow-up. Most of the hyperkalemia seen in these patients is non-severe and additional work is needed on how the mild hyperkalemia affects the course of CKD.