The use of immunotherapy agents is expanding in oncology patients. These medications, known as checkpoint inhibitors, promote patient T-cell based immune responses against malignancy. The currently available classes include PD-1 inhibitors, PDL-1 inhibitors and CTLA4 inhibitors. As the use of the immunotherapy agents is increased, more knowledge of adverse reactions is gained. Immune related adverse events in multiple organ systems may occur. The most common toxicities associated with checkpoint inhibitors include inflammatory responses in skin, gastrointestinal tract, lungs, endocrine organs, kidney and liver [1]. We present a case of PD-1 inhibitor nivolumab immune related adverse reaction causing hypophysitis, pleural and pericardial effusions.
An 81-year-old male with hepatocellular carcinoma metastatic to lungs and bone, who is receiving salvage therapy with nivolumab presents to the hospital with nausea, vomiting, weakness and altered mental status. Medical history includes peptic ulcer disease, glaucoma, benign prostatic hyperplasia, chronic hepatitis C infection treated with interferon and ribavirin, metastatic hepatocellular carcinoma diagnosed 2 years previously. Since diagnosis, he received trans-arterial chemoembolization, liver segmentectomy and radiation therapy for bony metastases. Five months after radiation therapy, he was started on salvage nivolumab biweekly. He is presenting one year after starting nivolumab. On initial evaluation, testing was significant for hyponatremia and elevated transaminases. Chest X-ray showed bilateral pleural effusions with suspected cardiomegaly (Figure 1). Echocardiogram demonstrated moderate sized anterior pericardial effusion with fibrinous exudate. He received fluid resuscitation and was started on empiric antibiotic therapy for infection.
 Figure 1: Chest X-ray showed bilateral pleural effusions with suspected cardiomegaly.
View Figure 1
Figure 1: Chest X-ray showed bilateral pleural effusions with suspected cardiomegaly.
View Figure 1
A diagnostic and therapeutic thoracentesis was performed, with 800 mL of yellow fluid removed from the right pleural space. Fluid studies were consistent with exudative effusion, based on pleural Lactate dehydrogenase 88, two thirds of serum 136. Cell counts indicated neutrophil predominance 59%. No malignant cells were identified on cytology. Pleural fluid gram stain and cultures were negative for bacteria. Blood and urine cultures were also negative for infection. Evaluation of his hyponatremia demonstrated panhypopituitarism. In consultation with Oncology, he was started on 1 mg/kg methylprednisolone for an immune adverse reaction to nivolumab. He clinically improved, and corticosteroids were tapered over 1 month. He continued to require hydrocortisone. Follow up imaging indicated resolution of his effusions (Figure 2).
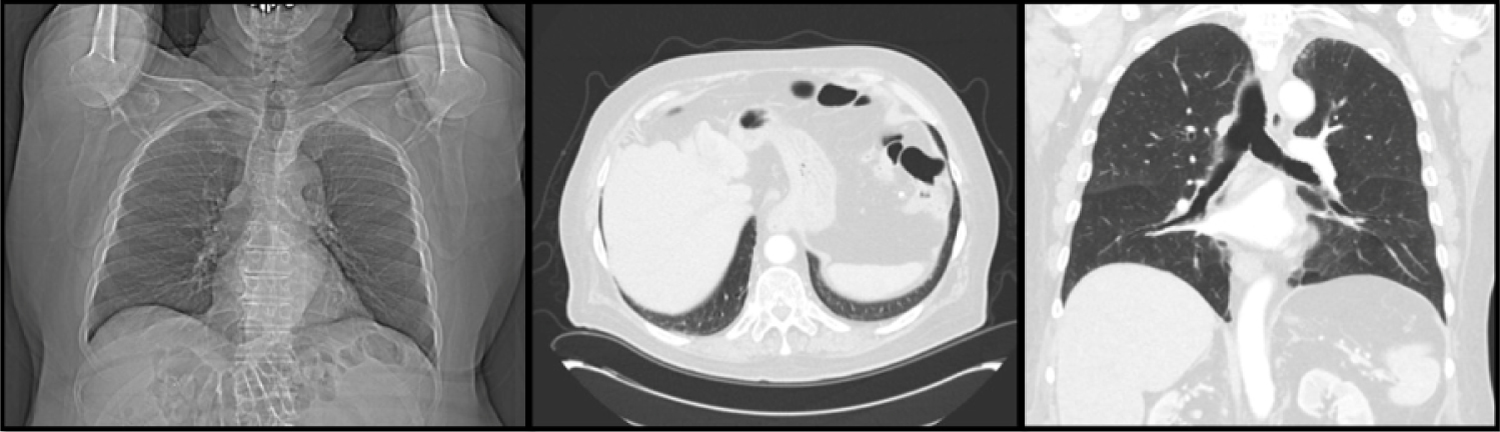 Figure 2: Image indicated resolution of patient effusions.
View Figure 2
Figure 2: Image indicated resolution of patient effusions.
View Figure 2
The case presented demonstrates multiple immune related adverse reactions to the checkpoint inhibitor PD-1, nivolumab. The use of nivolumab, initially indicated in melanoma, has been expanded to include use as a second line agent in hepatocellular carcinoma [2]. The most common pulmonary toxicity associated with nivolumab is pneumonitis [3]. Pleural effusions with nivolumab use have been documented [4]. Pericardial effusions and pericardial tamponade have been observed in several case reports of nivolumab use [5-7]. The combination of pleural and pericardial effusions has been noted, however these were malignant effusions [8]. The incidence of hypophysitis is estimated at 0.5% with PD-1 inhibitor use [3]. Increased incidence of hypophysitis is associated with higher dosing or concurrent use with CTLA-4 inhibitors [9]. The combination of multiple immune related adverse reactions is a unique feature of this case, previously shown on autopsy [10]. As the use of immunotherapy continues to expand, especially in those patients who are ineligible for chemotherapy, it is important to consider the potential adverse effects of these medications.