The demographic distribution, symptom profile and treatment outcomes in Rheumatoid arthritis associated interstitial lung disease (RA-ILD) has not been well described in South Africa due to a paucity of data.
The aim of the study was to determine the demographic profile of the disease, symptom and radiological presentation, as well as treatments used and their outcomes at Inkosi Albert Luthuli Central Hospital (IALCH), a specialised public government hospital treating the majority of patients with RA-ILD in the province of KwaZulu Natal, South Africa.
This was a retrospective electronic chart review of 61 patients who were diagnosed with RA-ILD between January 2010 and December 2015 at IALCH pulmonology clinic. Demographic data, symptom presentation, Pulmonary function testing (PFT), High resolution computerised tomography (HRCT) features and treatment modalities were analysed as well as outcome based on symptoms, PFT and HRCT.
There were 61 subjects, the majority being female (90.2%). Approximately 86.9% were 50 years and older. The majority of the subjects were ethnic Indian 72.1% (n = 44), followed by Black Africans 23% (n = 14) and then Whites 4.9% (n = 3). All patients were HIV negative. 90% of patients had a restrictive lung function test. The commonest HRCT pattern was a usual interstitial pneumonia. Patients treated with a combination of prednisone and azathioprine had a decline in forced vital capacity (p value 0.04). There was no improvement or deterioration in patients treated with either drug alone.
The management of RA-ILD is still a challenge. The combination of azathioprine and prednisone did not arrest disease progression in our study while either agent alone did not improve clinical and lung function parameters. Large randomised control studies are needed to further elucidate optimal management.
Rheumatoid arthritis, Interstitial lung disease, South Africa, Demographics, Treatment outcomes
Rheumatoid arthritis (RA) is a multi system inflammatory autoimmune disease affecting approximately 1% of the general population [1]. Interstitial lung disease (ILD) is one of the commonest primary pulmonary manifestations of RA [2]. Rheumatoid arthritis is commoner in females; however males are more affected in RA-ILD with a male to female ratio of 2:1 in some studies [3]. The onset of RA-ILD typically occurs in the fifth decade of life [3,4].
Age and smoking have been shown to be risk factors for the development of ILD [4] and high titres of Rheumatoid factor (RF) and anti cyclic citrullinated peptides (anti-CCP) are known risk factors for extra-articular manifestations of rheumatoid arthritis, including ILD [3-5]. Most patients with RA-ILD present with symptoms of dyspnoea or cough [3,6].
A restrictive pattern on Pulmonary function testing (PFT) is the most common pattern noted on spirometry in the majority of patients with RA-ILD, with or without a decrease in diffusing capacity of the lung for carbon monoxide (DLCO) [7]. An obstructive pattern may coexist on PFT and can be seen in patients manifesting airway involvement, such as Bronchiolitis obliterans (BO) [3].
In RA a variety of patterns are seen on High resolution computerised tomography (HRCT) scans, with the most common being Usual interstitial pneumonia (UIP), which occurs in 40-62% of cases. Non-specific interstitial pneumonia (NSIP) is the second most common pattern, occurring in approximately 11-32% of patients. Other types of patterns seen in RA-ILD include organising pneumonia, diffuse alveolar damage (DAD), Lymphocytic interstitial pneumonia (LIP) and Desquamative interstitial pneumonia (DIP). However these are less frequent [3].
Once a diagnosis of RA-ILD has been confirmed, treatment is generally directed to control the systemic disease with immunosuppressive agents while tailoring therapy to the underlying histopathological subtype [8,9].
Currently, there have not been enough randomised controlled trials comparing medications for the treatment of RA-ILD [8]. Corticosteroids are still the mainstay of therapy, particularly for cases of NSIP, organizing pneumonia or acute flare up of UIP [3,10]. Cyclophosphamide, azathioprine and cyclosporine have been used with varying digress of success [3].
There is a paucity of literature regarding RA-ILD in South Africa, its demographic distribution, clinical and radiological presentation, treatment outcomes as well as its relationship to HIV as the country has a high HIV burden. The aim of this study was to review the demographic data; PFT and HRCT scan pattern and treatment outcomes at a tertiary hospital in Durban, KwaZulu Natal, South Africa.
This was a retrospective electronic chart review. Patients 18 years and older referred to Inkosi Albert Luthuli Central Hospital pulmonology clinic diagnosed with RA-ILD between January 2010 and December 2015 were included in the study.
Demographic and clinical data, as well as data from special investigations, were captured retrospectively from medical records. Demographic data included age, gender and race. Clinical data captured were presenting symptoms, pre-existing medical conditions, current functional class, date of onset of initial symptoms, and treatment. Special investigations captured included results of HRCT, rheumatoid factor level, anti-CCP levels and PFT.
Ethical approval was granted by the University of KwaZulu Natal's Human Research Ethics Committee (ref. no. E044/2018).
Demographic data with a normal distribution such as age, gender and race are reported as means with Standard deviation (SD). Fisher's exact test was used for categorical variables. Statistical significance was regarded as a p-value < 0.05 unless otherwise stated. Continuous variables are presented as mean SD.
Change in symptoms between initial visit and last visit for cough, dyspnoea, Forced expiratory volume in 1 second (FEV1), or Forced vital capacity (FVC), was classified as worse or improved/static. Chi square or Fisher's exact test was used to compare the change by categorical data such as treatment. Initial mean FEV1 and FVC were compared by categories of RF and anti CCP using Mann-Whitney tests. Changes in FEV1 and FVC were coded as worse if the percentage difference change was 10% or more. Changes by treatment medications were analysed using Fisher's exact test. Pair wise comparisons were then done using azathioprine as a reference category using Fisher's exact tests. Data was analysed using Stata V13 statistical software.
There were 61 subjects who met the inclusion criteria with the majority being female by far at 90.2%. The majority of patients were above the age of 50 (86.9%). Most of the subjects were ethnic Indian 72.1% (n = 44), followed by Black Africans 23% (n = 14) and then Whites 4.9% (n = 3) (Table 1).
Table 1: Demographic details of the study population. View Table 1
Presenting symptoms were recorded at initial visit. The majority of patients had exertional dyspnoea as one of their initial symptoms followed by chronic cough. The New York Heart Association (NYHA) functional class at enrolment was recorded and 8 patients (13.1%) were class I disabled, 29 (47.5%) class II, 3 (3.3%) class III and none class IV. 22 patients (36.1%) had no dyspnoea at initial visit.
FEV1 and FVC were recorded in 58 patients with 3 patients having missing data. The median FEV1 was 1.54 L with IQR (0.87-2.34) SD 0. The median FVC was 1.87 L (0.75-3.02) IQR SD 0.52.
The majority of patients in our study had a restrictive pattern, 6 patients (9.8%) presented with obstructive pattern on PFT and 3 patients had missing data.
A transfer factor for carbon monoxide was recorded for some patients but the majority were not recorded due to lack of data.
HRCT analysis showed the following RA-ILD patterns: UIP 36.1% (n = 22), followed by NSIP pattern 19.7% (n = 12), bronchiolitis obliterans pattern 9.8% (n = 6) and others 34.4% (n = 21) (Figure 1).
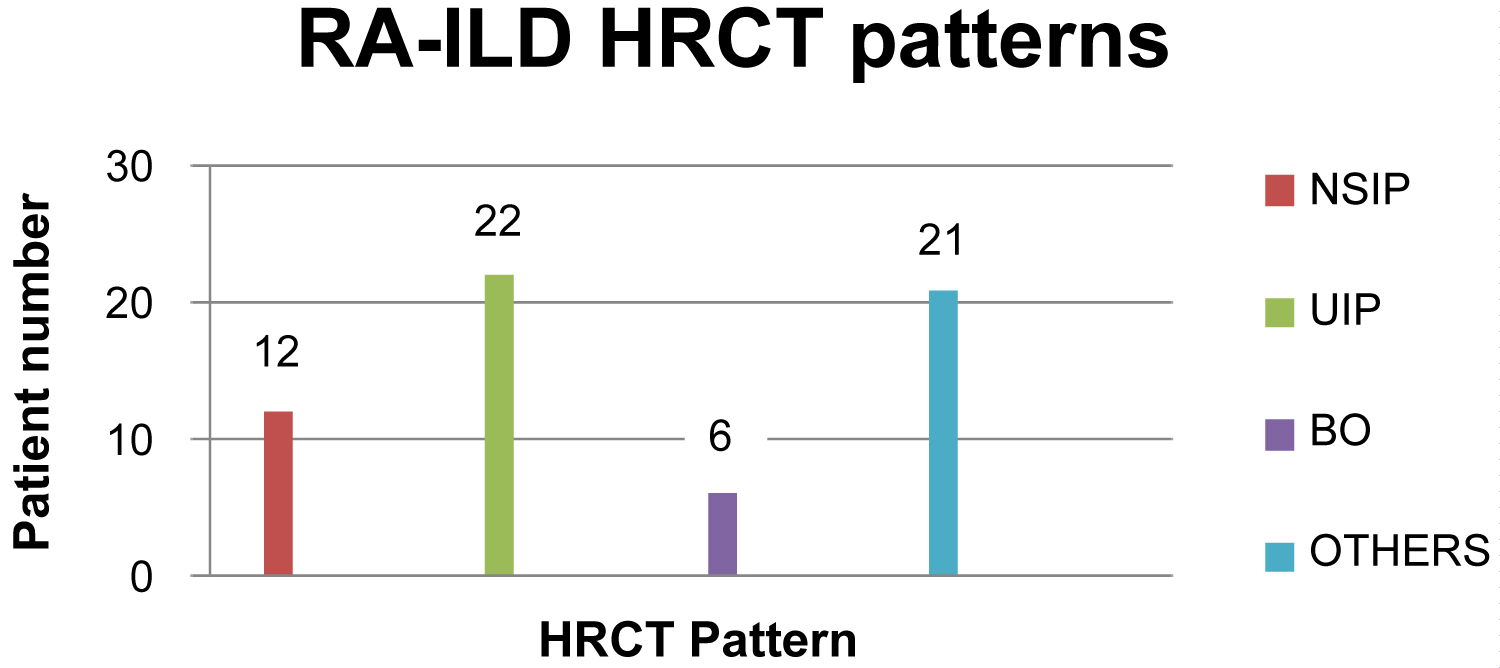 Figure 1: High resolution computed scan pattern.
View Figure 1
Figure 1: High resolution computed scan pattern.
View Figure 1
Thirty five patients (57.4%) had a positive anti-CCP titre with a median of 94 IU, (30.9-300). Forty one patients (67.2%) had an elevated RF antibody with a median of 116.5 IU, (46.5-207). There was no association between anti-CCP antibodies or RF antibodies with the presenting lung function or HRCT pattern.
Therapy was categorised into 4 groups: Azathioprine alone (n = 3, 4.9%), prednisone alone (n = 10, 16.4%), azathioprine combined with prednisone (n = 13, 21.3%), and no therapy (n = 35, 57.4%). Subjective responses with change in cough and dyspnoea were analysed before and after treatment as assessed by first and last visit. HRCT and PFT were used as objective responses to treatment before and after therapy.
The patients were classified as either disease progression or improved/static. There were no statistically significant differences in cough, dyspnoea, change in PFT or change in HRCT. However patients on prednisone and azathioprine combined had significant worsening of the FVC (p value 0.04).
All 61 patients were HIV negative.
RA-ILD is often subtle in onset, slowly progressive and of uncertain response to immune modulating agents [11]. This study aimed to clarify the current clinical, radiographic and pathologic status of RA-ILD and treatment and outcomes of patients seen at our quaternary hospital, IALCH, in Durban KwaZulu Natal. This is the main referral hospital in the province for public sector management of RA-ILD. KwaZulu Natal is the second most populous province in South Africa with a multi racial population comprising of 87% black Africans, 7.9% ethnic Indians and 3.9% white.
RA-ILD has been recognised as the most common extra-articular complication in RA patients. In this retrospective study, we evaluated ILD in RA patients referred to our clinic. We found that 86.9% of the patients (n = 53) with RA-ILD are between the ages of fifty to eighty years. About 13.1% of the patients (n = 8) were less than 50 years of age. This is similar to findings in most studies [3,12]. The likely explanation for the higher proportion of patients above age of 50 with ILD may be due to long duration of disease in the majority of RA patients or late diagnosis. Most patients were females (90.2%) as reported in many studies. The study done by Gabriel, et al., states that RA occurs 2-3 times more commonly in women with onset generally in adulthood, peaking in the 4th and 5th decade [13]. However male gender has been shown to be a risk factor for RA-ILD in a number of studies [12]. This gender imbalance has been observed across different ethnic populations, as well as in familial studies [12]. In our study we found that most of the patients are ethnic Indian followed by black African and then White. This finding in is not in line with the demographics of KwaZulu Natal and South Africa in which Black Africans comprise more than 80% of the total population of both the province and the country. It may be that the ethnic Indian race group may be more prone to ILD due to RA compared to black Africans or more prone to RA as a disease. Referral patterns in the different race groups may also be a factor.
Of particular interest are the differential associations observed between immunodeficiency secondary to HIV and RA-ILD, which we were unable to establish in our cohort. All our patients were HIV negative. Several reports have suggested that patients with established rheumatoid arthritis experience clinical improvement after the development of immunodeficiency secondary to HIV [14]. Tarr, et al. observed that most HIV positive RA patients in their cohort had lower joint counts and composite disease activity scores despite stopping immune modulating therapy compared to HIV negative controls, supporting the suggestion that HIV infection improves RA disease activity [15]. There have been no studies thus far in South Africa addressing the correlation between HIV and RA-ILD activity.
The majority of patients in our study had a restrictive pattern on PFT which is similar to most studies [3,6]. We identified 6 patients (9.8%) who presented with an obstructive pattern.
The present study confirms current knowledge from other parts of the world: For example Silva, et al. showed that the predominant pattern of ILD in RA is UIP [14]. In our study UIP accounted for more than one-third of patients (36.1%, n = 22), followed by NSIP pattern (19.7%, n = 12), bronchiolitis obliterans (9.8%, n = 6) and others (34.4%, n = 21). The UIP pattern is independently associated with a poorer prognosis and an increase in all-cause mortality [11].
The differential associations observed between anti-CCP antibodies and rheumatoid factor in relation to initial symptoms such as cough, dyspnoea, initial PFT, HRCT outcome and HRCT abnormalities are of particular interest. Anti-CCP antibodies are highly specific for RA and are associated with poor articular prognosis [16]. Consistent with most other studies in RA, we have not identified a relationship between anti-CCP antibodies or rheumatoid factor and initial symptoms such as cough and dyspnoea or initial PFT, nor HRCT patterns. However, a study from New Zealand demonstrated an association between anti-CCP antibodies and both physiologic abnormalities and bronchial wall thickening on HRCT [16]. This may suggest an association between the antibody and the interstitial lung disease.
The management of patients with RA-ILD remains unclear. It is not known whether treatment will delay or stop progression of the disease activity. The mainstay of treatment in RA-ILD is immune suppressive agents. First line therapy is steroids such as prednisone for prolonged duration especially for patterns such as NSIP and OP. In this retrospective study we found 16.4% of patients had been treated with prednisone alone, 4.9% with azathioprine alone, and 21.1% with both prednisone and azathioprine. More than half the patients (57.4%) had no specific treatment for the RA-ILD as they had irreversible disease based on HRCT. There were no differences in symptoms or PFT outcome between first and last visit. However we found that, patients who were on both prednisone and azathioprine had a significant decline in FVC. This is consistent with the PANTHER study which revealed an increased rate of death and hospitalisation in the combination therapy group with prednisone and azathioprine compared with placebo in patients with Idiopathic pulmonary fibrosis (IPF) [17]. It is likely that patients with a UIP pattern RA- LD follow the same outcome as those with IPF and those patients with UIP pattern in RA-ILD have a worse prognosis.
Several studies have raised the possibility that the response to therapy correlates with the histological patterns in RA-ILD. RA-ILD with the NSIP pattern has a more favourable response to therapy than that with the UIP pattern, resulting in a better prognosis. Patients with the OP pattern typically show rapid improvements in response to corticosteroid therapy [18].
This was a retrospective review of electronic data and only data captured and available could be analysed. It was a small sample size at the main centre in our province. The small sample size as well as missing data may have biased any associations. However, RA ILD is a disease with a very low prevalence; hence smaller sample sizes are expected. There is the possibility of overestimation as the study was conducted in a specialised clinic in the major referral hospital for the province, which may have resulted in referral bias. There may also have been patients with interstitial lung disease, who may have been missed because they were asymptomatic.
This is the first study to our knowledge, in South Africa, to study the demographics and treatment of RA-ILD in our multi racial society. The majority of patients with RA-ILD at IALCH are of Indian descent, contrary to the population demographics of the country which is greater than 80% Black African. None of our patients were HIV infected which may attest to previous reports of a protective effect of HIV on RA. Active treatment of RA-ILD with prednisone, azathioprine or both combinations appears to have had no positive impact on symptoms or PFT. However, these results must be interpreted with caution in light of this being a retrospective study with its significant shortcomings.
Further prospective multi-centre randomised controlled trials are required in this field to determine the optimal drug treatment to manage RA-ILD as well as the impact of HIV infection.