International Journal of Respiratory and Pulmonary Medicine
Preliminary Study of Expression of KL-6, FGF-10 and MMP-9 in the Primary Spontaneous Pneumothorax
Jinxin Cao, Jinbai Miao and Hui Li*
Department of Thoracic Surgery, Beijing Chao-yang Hospital, Capital Medical University, P.R. China
*Corresponding author: Hui Li, Department of Thoracic Surgery, Beijing Chao-yang Hospital, Capital Medical University, Beijing 100020, P.R. China, Tel: 8610-85231165, E-mail: huilee@vip.sina.com
Int J Respir Pulm Med, IJRPM-2-018, (Volume 2, Issue 2), Research Article; ISSN: 2378-3516
Received: March 04, 2015 | Accepted: March 30, 2015 | Published: April 03, 2015
Citation: Cao J, Miao J, Li H (2015) Preliminary Study of Expression of KL-6, FGF-10 and MMP-9 in the Primary Spontaneous Pneumothorax. Int J Respir Pulm Med 2:018. 10.23937/2378-3516/1410018
Copyright: © 2015 Cao J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Pathogenesis of primary spontaneous pneumothorax (PSP) is unclear and has been rarely reported in the literature. Thus, we measured expression of Krebs Vonden Lungen-6 (KL-6), fibroblast growth factor-10 (FGF-10), and matrix metalloproteinase-9 (MMP-9) in PSP samples and evaluated their significance on the development and progress of the disease.
Methods: Immunohistochemical staining and ELISA were used to measure expression of KL-6, FGF-10, and MMP-9 in the pulmonary bullae and the peripheral normal lung tissue in 24 cases of PSP. Statistical results were analyzed using the Student�s paired t-test.
Results: Immunohistochemical data revealed that KL-6 and FGF-10 expression in the pulmonary bullae was significantly higher than in peripheral normal lung tissue, but MMP-9 expression did not differ. ELISA confirmed that KL-6 and FGF-10 expression in the pulmonary bullae was significantly higher than the peripheral normal lung tissue (P < 0.05); but MMP-9 expression again did not differ (P >0.05).
Conclusion: Expression of KL-6 and FGF-10 in pulmonary bullae of PSP patients was significantly higher than in the peripheral normal lung tissue, suggesting that KL-6-mediated pulmonary fibrosis and abnormal FGF-10 expression-induced congenital abnormalities in development of bronchi may be associated with PSP. MMP-9 was not different in both groups, suggesting that further correlation between MMP-9-mediated inflammation and PSP remains unclear.
Keywords
Primary spontaneous pneumothorax, Krebs Vonden Lungen-6(KL-6), Fibroblast growth factor-10 (FGF-10), Mmatrix metalloproteinase-9 (MMP-9)
Introduction
Primary spontaneous pneumothorax (PSP) refers to a fracture of the pulmonary parenchyma or visceral pleura, resulting in gas accumulation in the pleural cavity and this may be not be accompanied by underlying lung diseases [1]. PSP is commonly treated with emergency thoracic surgery. The PSP incidence is 7.4-18 of 10,000 persons annually for men and 1.2-6 of 10,000 persons for females [2]. PSP occurs in tall, slender individuals who are male, smoking, and have fibrosis as detected by a chest X-ray. Most PSP symptoms are mild and patients do not seek medical treatment. Thus, the actual PSP incidence may be greater than seen with clinical findings.
Pathogenesis of PSP remains unclear and may arise from terminal airway inflammation [3], genetic factors [4], abnormal fibrous connective tissue [5], abnormal anatomy of the bronchial tree [6], emaciation with large negative intrapleural pressure [7], insufficient blood supply to the apex, low body mass index, and caloric restriction [8], as well as elevated plasma concentrations of aluminum [9]. Pulmonary fibrosis, congenital dysplasia, and inflammation have been thought to be responsible for PSP pathogenesis but little data support these assertions. We measured 3 cytokines (i.e., Krebs Vonden Lungen-6 (KL-6), fibroblast growth factor-10 (FGF-10), and matrix metalloproteinase-9 (MMP-9)) to learn whether they contributed to pulmonary fibrosis, congenital abnormalities, and inflammation and we correlated these data with PSP findings.
Materials and Methods
Sample sources
A total of 24 patients (18 males; age range 13�25 years, mean age=17 years) were included and underwent thoracoscopic resection of pulmonary bullae in our hospital from January 1, 2012 to June 30, 2012 due to PSP. All patients had no history of smoking or other comorbidities (such as pulmonary fibrosis or inflammation). Pneumothorax and/or the presence of bullae were confirmed in all patients by chest X-ray or chest computed tomography prior to surgery. Intraoperational pulmonary bullae were confirmed in all patients. Pathological thoracoscopic examination of resected pulmonary bullae was performed and tissue samples were chosen for the experimental group. Controls included normal peripheral lung tissues. All tissues were stored at -80�C.
Reagents
Mouse anti-human KL-6 monoclonal antibody, rabbit anti-human FGF-10 polyclonal antibody, and rabbit anti-human MMP-9 polyclonal antibody were purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX). Goat anti-mouse/rabbit secondary antibodies and 3,3�- diaminobenzidine (DAB) enhanced liquid substrate kit was purchased from (Beijing Zhong Shan � Golden Bridge Biological Technology Co., Ltd., Beijing, China). KL-6, FGF-10, and MMP-9 enzyme-linked quantitative detection kits were purchased from Shanghai Senxiong Technology Industry Co., Ltd., Shanghai, China). Total protein quantitative detection kit was purchased from Pierce Biotechnology., Inc. (BCA protein assay kit, Thermo Fisher Scientific, Waltham, MA).
Immunohistochemical staining
Tissue samples were post-fixed in formalin solution, followed by paraffin embedding (tissue sections 5�m thickness). All tissue sections were incubated in the oven for 45 min, followed by hydrating through an ethanol gradient and ultimately into water. EDTA (pH 8.0) solution was used to heat retrieve the antigen for 18 min, followed by cooling at room temperature and washing. A 3% hydrogen peroxide solution was used to quench endogenous peroxidase. After washing PBS in 2 times, 1:50 dilutions of KL-6, FGF-10, and MMP-1 primary antibodies were added to individual tissue sections and incubated at room temperature for 1 h, followed by PBS washing 3 times. Secondary antibodies were then added to tissue sections and incubated at room temperature for 15 min, followed by PBS washing 3 times. DAB enhanced liquid substrate was used for color development which was terminated by rinsing with water. Tissue sections were counterstained with hematoxylin dye and differentiated with hydrochloric acid alcohol. Ammonium hydroxide solution was used convert hematoxylin staining into a deep purplish blue color. Tissue sections were dehydrated in an ethanol gradient, followed by xylene clearance before mounting with neutral gum. Immunohistochemical staining was examined under light microscopy.
ELISA
Tissue samples from the experimental and control groups were thawed on a filter paper to absorb blood and water, weighed, and to each was added 1.5 ml PBS and phenylmethanesulfonylfluoride (PMSF, protease inhibitor) mixture. Tissue homogenization was followed by centrifugation at 4�C to collect the supernatant. In a 96-well plate, precoated with KL-6, FGF-10, or MMP-9 antibody, 100μl of antigen standard or sample supernatant was added per well as detailed in the ELISA kit manual. The 96-well plate was incubated at 37�C for 120 min. After thorough washing, 50μl anti-primary antibody working solution was added into each well and incubated at 37�C for 60 min. After thorough washing, 100μl enzyme-conjugated antibodies was added and incubated at 37�C for 60 min. After thorough washing, 100μl substrate solution was incubated at 37�C for 10 min, followed by reaction termination with 50μl stop solution. Absorbance (optical density, OD) was measured at 450nm. Standard calibration curves were used to estimate of KL-6, FGF-10, and MMP-9 in the samples of both experimental and control groups. Each cytokine was then quantified by dividing the amount of total protein used in each ELISA sample.
Statistical analysis
SPSS17.0 software was used for statistical analysis and the Student�s paired t-test was used to analyze data between experimental and control groups. P< 0.05 was considered to be statistically significant.
Results
Experimental results of KL-6
Expression of KL-6 in the experimental group (Figure 1A) was significantly higher than in controls (Figure 1B). Both groups were normally distributed. KL-6 mean concentration was 0.15 ± 0.10 in the experimental group and 0.06 ± 0.02 in the control group. KL-6 in the experimental group was significantly higher than in controls (t = 2.320, P< 0.05).
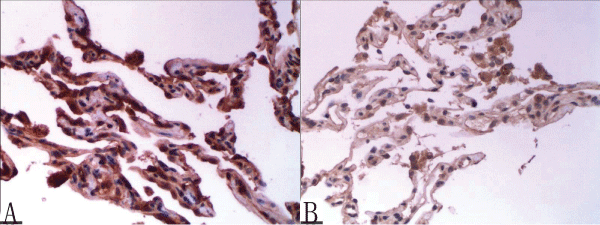
Figure 1: Representative image of KL-6 IHC staining in the experimental group (positive KL-6 expression is brown; hematoxylin counterstained nuclei are blue)
(A) KL-6 expression in the experimental group and (B) in the control group (�200 magnification)
View Figure 1
Experimental results of FGF-10
Expression of FGF-10 in the experimental group (Figure 2A) was significantly higher than in the control group (Figure 2B). FGF-10 mean concentration was 0.04 ± 0.03 in the experimental group and 0.01 ± 0.00 in the control group. FGF-10 in the experimental group was significantly higher than in the control group (t=2.624, P < 0.05).
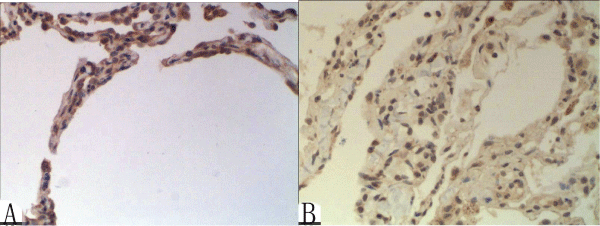
Figure 2: Representative image of FGF-10 IHC staining in the experimental group (positive FGF-10 expression is brown; hematoxylin counterstained nuclei are blue)
(A) FGF-10 expression in the experimental group and (B) in the control group (�200 magnification)
View Figure 2
Experimental results of MMP-9
MMP-9 expression did not differ between groups (Figures 3A,3B) MMP-9 mean concentration was 0.003± 0.003 in the experimental group and 0.001 ± 0.00 in the control group.
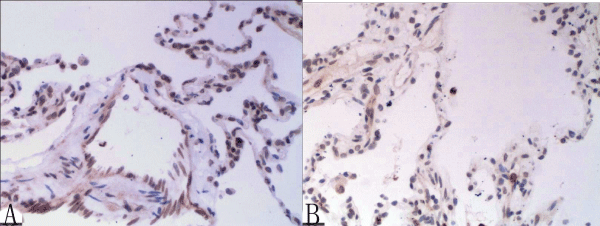
Figure 3: Representative image of MMP-9 IHC staining in the experimental group (positive MMP-9 expression is brown; hematoxylin counterstained nuclei are blue)
(A) MMP-9 expression in the experimental group and at (B) in the control group (�200 magnification)
View Figure 3
Discussion
KL-6, discovered by Kohno and colleagues in 1985 is a member of the mucin protein (MUC1) family, and expressed to a certain extent in normal alveolar type II cells and terminal bronchial epithelial cells. KL-6 is a fibroblast chemoattractant that induces fibroblasts migration and aggregation in damaged alveolar space to mediate alveolar fibrosis, KL-6 may also be involved in the formation and development of PSP. Proliferation of fibrous connective tissue leads to alveolar damage which causes scar formation. Scars reduce alveolar elasticity compromising alveolar gas exchange and ultimately forming pulmonary bullae.
KL-6 is a sensitive biomarker of interstitial lung diseases (e.g., idiopathic pulmonary fibrosis, hypersensitivity pneumonitis, and radiation pneumonitis) due to its role in pulmonary fibrosis. Satoh�s group reported that in 219 cases (i.e., 152 cases of idiopathic pulmonary fibrosis and 67 cases of pulmonary fibrosis-related collagen disease) for the experimental group and 22 cases (i.e., 13 cases of chronic obstructive pulmonary disease and 9 cases of bronchial asthma) in the control group, that KL-6 expression in the experimental group was significantly higher than the control group. Greater KL-6 expression is associated with poorer prognosis and high incidences of respiratory failure and mortality [10]. Kruit�s group collected 242 cases of interstitial lung disease (experimental group) and 327 healthy volunteers (control group) and reported that KL-6 expression in the experimental group was significantly higher than in the control group [11]. Thus, among the diffuse pulmonary parenchyma, alveolar inflammation, and interstitial fibrosis as the pathological bases of interstitial pulmonary disease, KL-6 may be key to pulmonary fibrosis. We report that KL-6 expression in lung tissues of PSP patients was significantly higher than in the peripheral normal lung tissue (controls), suggesting that PSP lesions may have KL-6-mediated pulmonary fibrosis as was observed in interstitial lung disease.
FGF-10 is a member of the fibroblast growth factor family and can stimulate epithelial cell growth. Through the induction and regulation of growth and mitosis of epithelial cells in the lung, FGF-10 is mitogenic and a chemoattractant [12]. Lung epithelial cells are grown toward the aggregation of FGF-10 to form embryos and continue growing [13]. FGF-10 expression in abnormal tissue induces overgrowth of distal tubules and expansion of epithelial tubules, resulting in developmental defects in different tissue areas, leading to the development of pulmonary bullae.
FGF-10 is necessary for lung-bronchial growth and development. Kim�s group used transgenic technology to knock out upstream regulatory factors of FGF-10 in a mouse model. They reported impairment of lung-bronchial development with emphysema-like changes in adult mice [14]. Perl�s group used doxycycline inducible system and measured expression of antagonistic factor Sprouty-4 (Spry-4) of FGF-10 receptor in mouse embryonic lung epithelial cells (a model or severe pulmonary hypoplasia) [15]. These reports support that abnormal FGF-10 expression leads to abnormal lung-bronchial growth and development. We report that FGF-10 expression in PSP lung lesions was significantly higher than in controls, suggesting that PSP lesions also had abnormal expression of FGF-10 which caused abnormal lung-bronchial growth and development.
MMP-9 is a zinc and calcium dependent matrix protein hydrolase of the matrix metalloproteinase family. MMP-9 can degrade extracellular matrix components in diverse proteins during inflammation, tissue remodeling, pathophysiological cell migration, angiogenesis, and other processes. Through protein aggregation, MMP-9 directs activity of inflammatory cells in the airway lumen and wall, worsening local bronchial inflammatory responses and causing airflow obstruction. In contrast, in response to inflammatory cytokines, MMP-9 expands the alveolar cavity and weakens alveolar elasticity via damaging alveolar matrix components, resulting in gas retention. Expression of MMP-9 in normal tissue is minimal; whereas MMP-9 expression is increased via stimulation of inflammatory cytokines.
MMP-9 is a marker for chronic obstructive pulmonary disease (COPD). Simpson�s group collected 100 cases of COPD (experimental group) and 61 healthy volunteers (control group) and measured sputum MMP-9. They report that MMP-9 in the experimental group was significantly higher than in controls (P < 0.05) [16]. Chen and co-workers reported in a meta-analysis derived from Pubmed and the CNKI database (n = 14 for each experimental and control groups) that there was a significant correlation between MMP-9 expression and the risk of COPD in healthy smokers as controls [17]. Thus, in COPD, with pathological features of chronic inflammation including airway inflammation, pulmonary parenchyma, and pulmonary vessels, MMP-9 may be important in the inflammatory process. Our data did not confirm these findings, however. This may be explained by the fact that PSP inflammation is not mediated by MMP-9 or that the limited occurrence and extent of PSP inflammatory responses led to non-significant differences of MMP-9 expression between the experimental and control groups. Also, our sample was small, and we may require more subjects to observe significant differences in MMP-9.
At this time, pathogenesis of PSP is not entirely clear. We report that KL-6 mediated pulmonary fibrosis and abnormal expression of FGF-10 which led to lung-bronchial congenital abnormalities that may be associated with the incidence of PSP. Our work offers a theoretical basis for the study of PSP pathogenesis. KL-6 and FGF-10 are important markers for interstitial lung disease and asthma via their roles in pulmonary fibrosis and lung development. Therefore, confirmation of the correlation of pathogenesis of PSP and interstitial lung disease and asthma are needed to predict PSP development. We confirmed high expression of KL-6 and FGF-10 in pulmonary bullae but confirmation of differences in serum KL-6 and FGF-10 in PSP patients is needed. To date, PSP diagnosis is made by chest X-ray and CT. Conformation of cytokine expression may help with early diagnosis and prevention of PSP.
References
-
Cheng G, Zhang ZY (2008) The etiology of spontaneous pneumothorax. Journal of Clinical Pulmonary Medicine 13: 322-324.
-
Bense L, Eklund G, Wiman LG (1987) Smoking and the increased risk of contracting spontaneous pneumothorax. Chest 92: 1009-1012.
-
Schramel F, Meyer CJ, Postmus PE (1995) Inflammation as a cause of spontaneous pneumothorax and emphysema-like changes: results of bronchoalveolar lavage. Eur Respir J 8: 397.
-
Morrison PJ, Lowry RC, Nevin NC (1998) Familial primary spontaneous pneumothorax consistent with true autosomal dominant inheritance. Thorax 53: 151-152.
-
Loeys BL, Matthys DM, de Paepe AM (2003) Genetic fibrillinopathies: new insights in molecular diagnosis and clinical management. Acta Clin Belg 58: 3-11.
-
Bense L, Eklund G, Wiman LG (1992) Bilateral bronchial anomaly. A pathogenetic factor in spontaneous pneumothorax. Am Rev Respir Dis 146: 513-516.
-
Fujino S, Inoue S, Tezuka N, Hanaoka J, Sawai S, et al. (1999) Physical development of surgically treated patients with primary spontaneous pneumothorax. Chest 116: 899-902.
-
Coxson HO, Chan IH, Mayo JR, Hlynsky J, Nakano Y, et al. (2004) Early emphysema in patients with anorexia nervosa. Am J Respir Crit Care Med 170: 748-752.
-
Leo F, Venissac N, Drici MD, Mouroux J (2005) Aluminium and primary spontaneous pneumothorax. A suggestive but unconfirmed hypothesis. Interact Cardiovasc Thorac Surg 4: 21-22.
-
Satoh H, Kurishima K, Ishikawa H, Ohtsuka M (2006) Increased levels of KL-6 and subsequent mortality in patients with interstitial lung diseases. J Intern Med 260: 429-434.
-
Kruit A, Gerritsen WB, Pot N, Grutters JC, van den Bosch JM, et al. (2010) CA 15-3 as an alternative marker for KL-6 in fibrotic lung diseases. Sarcoidosis Vasc Diffuse Lung Dis 27: 138-146.
-
Peters K, Werner S, Liao X, Wert S, Whitsett J, et al. (1994) Targeted expression of a dominant negative FGF receptor blocks branching morphogenesis and epithelial differentiation of the mouse lung. EMBO J 13: 3296-3301.
-
Bellusci S, Furuta Y, Rush MG, Henderson R, Winnier G, et al. (1997) Involvement of Sonic hedgehog (Shh) in mouse embryonic lung growth and morphogenesis. Development 124: 53-63.
-
Kim N, Yamamoto H, Pauling MH, Lorizio W, Vu TH (2009) Ablation of lung epithelial cells deregulates FGF-10 expression and impairs lung branching morphogenesis. Anat Rec (Hoboken) 292: 123-130.
-
Perl AK, Hokuto I, Impagnatiello MA, Christofori G, Whitsett JA (2003) Temporal effects of Sprouty on lung morphogenesis. Dev Biol 258: 154-168.
-
Simpson JL, McDonald VM, Baines KJ, Oreo KM, Wang F, et al. (2013) Influence of age, past smoking, and disease severity on TLR2, neutrophilic inflammation, and MMP-9 levels in COPD. Mediators Inflamm 2013: 462934.
-
Chen L, Wang T, Liu L, Shen Y, Wan C, et al. (2013) Matrix metalloproteinase-9 -1562C/T promoter polymorphism confers risk for COPD: a meta-analysis. PLoS One 8: e60523.





