International Journal of Neurology and Neurotherapy
The Importance of Stimulation Cycle in Vagus Nerve Stimulation for Drug-Resistant Epilepsies- Our Experience and Literature Review
Nicola Montano*, Filomena Fuggetta, Fabio Papacci, Rina Di Bonaventura, Mario Meglio and Gabriella Colicchio
Institute of Neurosurgery, Catholic University School of Medicine of Rome, Italy
*Corresponding author:
Nicola Montano, Institute of Neurosurgery, Catholic University, Largo Agostino Gemelli, 800168 Rome, Italy, Tel: +39 0630154120,+39 0630154358; Fax: +39 063051343; E-mail: nicolamontanomd@yahoo.it
Int J Neurol Neurother, IJNN-1-013, (Volume 1, Issue 1), Review Article; ISSN: 2378-3001
Received: October 09, 2014 | Accepted: December 15, 2014 | Published: December 17, 2014
Citation: Montano N, Fuggetta F, Papacci F, Bonaventura RD, Meglio M, et al. (2014) The Importance of Stimulation Cycle in Vagus Nerve Stimulation for Drug-Resistant Epilepsies- Our Experience and Literature Review. Int J Neurol Neurother 1:013. 10.23937/2378-3001/1/1/1013
Copyright: © 2014 Montano N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The impact of stimulation cycle on the outcome of patients submitted to vagal nerve stimulation (VNS) has been only marginally investigated in the literature. Nonetheless this is an important factor in term of tolerability of side effects, duration of generator and costs of therapy. Here the role of this parameter was evaluated on 21 patients who underwent to VNS implant at our Institution from January 1994 to February 2011 and responded to VNS (they reported a stable decrease of seizure number at least of 50%). We studied 3 stimulation cycles: slow (30''on-5'off), intermediate (30''on-3'off), fast (7''on-20''off). Each cycle was 1 year in duration. The stimulation cycle was recorded at the latest available follow-up (FU) for each patient (mean FU was 80.42 ± 54.01 months). Our protocol was to start with the slow cycle, to switch to the fast cycle and then to the intermediate one. The mean seizure frequencies before VNS and at latest FU were 26.08 ± 29.73 and 7.54 ± 10.22, respectively (p< 0.0001; Wilcoxon Signed Rank Test). At latest FU, 11 cases (52.38%) were using the slow cycle and 8 (38.09%) the intermediate one. Among patient with a FU longer than 3 years (n=16), 15 of them (93.75%) were using slow or intermediate cycles. When considering the subgroup of patients with a 75% of reduction of number of crisis (n=12) nobody was using the fast cycle. Our results indicate that in patients responding to VNS the slow and intermediate cycles maintain the benefit of stimulation. If there is no response with these two cycles the probability of become a responder with the fast cycle is low.
Keywords
Vagus nerve stimulation, Epilepsy, Stimulation cycle
Introduction
Vagus nerve stimulation (VNS) is an adjunctive treatment for drug-resistant epileptic patients excluded from ablative surgery. The results reported in the literature are highly variable due to a widening of indications in the last years [1-4], the lack of defined prognostic factors [5] and the absence of full understanding of mechanism of action [6,7]. Recently we published on this topic and showed that the lesional etiology and an implant age less than 18 years were associated with an higher probability to be a responder after VNS [8]. Nevertheless the role of patient's stimulation cycle was not investigated in that analysis. Moreover this data is only marginally reported in the literature [2,9-11] and not investigated in a recent meta-analysis analyzing the results of this therapy in drug resistant epilepsies [5]. Recently an evidence-based guideline update on VNS failed to find recommendation for this topic [12]. Here we investigated how stimulation cycle affects the efficacy of VNS. We also reviewed the pertinent literature.
Methods
We retrospectively reviewed the clinical records and stimulation parameters of 21 patients (14 M and 7 F) (Table 1) submitted to VNS implant at the Functional and Spinal Neurosurgery of the Catholic University from January 1994 to February 2011. To minimize the impact of other possible confounding factors we included in this analysis only the patients who responded to VNS (we identify patients as "responders" when the decrease of seizure number was at least 50%). Pre-implant evaluation, surgical procedure and post-implant evaluation have previously been reported [13]. Mean follow-up (FU) was 80.42 ± 54.01 months. The stimulation frequency was 30 Hz and the pulse width 500 µsec. Stimulation cycles used were the following: slow (30''on-5'off), intermediate (30''on-3'off), fast (7''on-20''off). Each cycle was 1 year in duration. Our protocol was to start with the slow cycle, to switch to the fast cycle and then to the intermediate one. Ethical approval of this protocol was previously obtained by the institutional review board [13]. We recorded the stimulation cycle at the latest available FU for each patient.
![]()
Table 1: Clinical records and stimulation parameters of 21 "responders" to vagal nerve stimulation
View Table 1
Results
The mean age of onset of epilepsy was 9.22 ± 10.02 years (range: 1-54 years) and the mean pre-implant epilepsy duration was 21.71 ± 11.07 years (range: 3-42 years). The mean age at implant was 31.42 ± 14.7 years (range: 10-62 years). Etiology was non-lesional in 5 patients and lesional in 16 patients (cortical malformations n=2, ischemia n=8, tumor n=2, infection n=1 and tuberous sclerosis n=3). The mean seizure frequencies before VNS and at latest FU were 26.08 ± 29.73 and 7.54 ± 10.22, respectively (p< 0.0001; Wilcoxon Signed Rank Test) (Figure 1). At latest FU, 11 cases (52.38%) were using the slow cycle, 8 (38.09%) the intermediate one and only 2 patients (14.28%) the fast cycle. Among patients with a FU longer than 3 years (n=16), 15 of them (93.75%) were using slow or intermediate cycles. When considering the subgroup of patients who reached a 75% of reduction of seizures number (n=12) nobody was using the fast cycle.
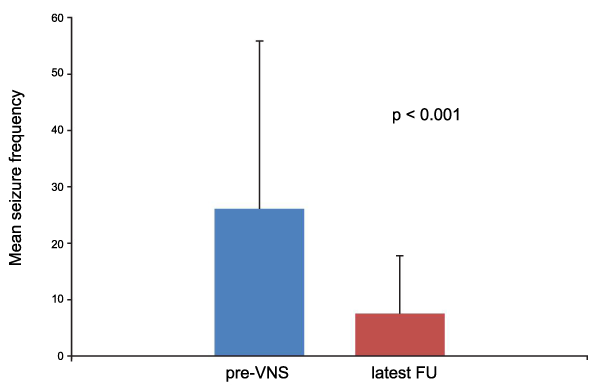
Figure 1: Wilcoxon Signed Rank Test.
View Figure 1
Discussion
Our results indicate that in patients "responders" to VNS the slow and intermediate cycles maintain the benefit of stimulation. We investigated this parameter only in the responder patients to avoid the interference of other possible confounding variables that influence the prognosis of these patients. We think that this observation is interesting because the stimulation cycle affects the duration of generator that is directly correlated to the cost of this therapy. When examining the literature we found that the latest published meta-analysis didn't take into account the role of this parameter [5] and that the evidence to support the use of a determined cycle to reduce seizure occurrence was found insufficient in a recent evidence-based guideline update [12]. Furthermore the role of stimulation cycle in VNS therapy was fully investigated only marginally [2,9-11]. Moreover these studies had a FU generally short (mean FU ranges from 3 to 15.8 months) (Table 2). While Labar [2] found that stimulation parameters did not affect seizure rates in their groups, Shermann and colleagues [9], evaluating two stimulation cycles (fast and slow cycles), evidenced as patients with stimulation-on period of 30 sec (slow cycle) had a significantly better seizure outcome than patients with stimulation-on periods of 7 sec (fast cycle). Moreover it has been showed that in the first 3 months of therapy, initial settings of 30 seconds on/3 minutes off are well tolerated, and produced the most 75% responders [11] and that patients in the settings of 30 sec on and 5 min off improve in their response over 1 year period [10]. In our study the mean FU was 80.42 ± 54.01 months and we found that in patients with a FU more than 3 years, all but one case were using the slow or intermediate cycle.
![]()
Table 2: Literature review of studies comparing stimulation cycles in vagus nerve stimulation for drug-resistant epilepsies
View Table 2
Conclusions
Our study has some limitations due to the limited number of patients and the retrospective nature of data. However based on our observations and the reported literature, in managing patients after VNS implantation, we suggest to start with the slow cycle and, if there is no response, to switch to intermediate one. At that stage the probability for the patient of being a responder with the fast cycle is very low. Obviously further studies that should be multicentric and randomized are needed.
References
-
Frost M, Gates J, Helmers SL, Wheless JW, Levisohn P, et al. (2001) Vagus nerve stimulation in children with refractory seizures associated with Lennox-Gastaut syndrome. Epilepsia 42: 1148-1152.
-
Labar D (2004) Vagus nerve stimulation for 1 year in 269 patients on unchanged antiepileptic drugs. Seizure 13: 392-398.
-
Parker AP, Polkey CE, Binnie CD, Madigan C, Ferrie CD, et al. (1999) Vagal nerve stimulation in epileptic encephalopathies. Pediatrics 103: 778-782.
-
Rychlicki F, Zamponi N, Trignani R, Ricciuti RA, Iacoangeli M, et al. (2006) Vagus nerve stimulation: clinical experience in drug-resistant pediatric epileptic patients. Seizure 15: 483-490.
-
Englot DJ, Chang EF, Auguste KI (2011) Vagus nerve stimulation for epilepsy: a meta-analysis of efficacy and predictors of response. J Neurosurg 115: 1248-1255.
-
Casazza M, Avanzini G, Ferroli P, Villani F, Broggi G (2006) Vagal nerve stimulation: relationship between outcome and electroclinical seizure pattern. Seizure 15: 198-207.
-
Helmers SL, Begnaud J, Cowley A, Corwin HM, Edwards JC, et al. (2012) Application of a computational model of vagus nerve stimulation. Acta Neurol Scand 126: 336-343.
-
Colicchio G, Montano N, Fuggetta F, Papacci F, Signorelli F, et al. (2012) Vagus nerve stimulation in drug-resistant epilepsies. Analysis of potential prognostic factors in a cohort of patients with long-term follow-up. Acta Neurochir (Wien) 154: 2237-2240.
-
Scherrmann J, Hoppe C, Kral T, Schramm J, Elger CE (2001) Vagus nerve stimulation: clinical experience in a large patient series. J Clin Neurophysiol 18: 408-414.
-
DeGiorgio CM, Thompson J, Lewis P, Arrambide S, Naritoku D, et al. (2001) Vagus nerve stimulation: analysis of device parameters in 154 patients during the long-term XE5 study. Epilepsia 42: 1017-1020.
-
DeGiorgio C, Heck C, Bunch S, Britton J, Green P, et al. (2005) Vagus nerve stimulation for epilepsy: randomized comparison of three stimulation paradigms. Neurology 65: 317-319.
-
Morris GL 3rd, Gloss D, Buchhalter J, Mack KJ, Nickels K, et al. (2013) Evidence-based guideline update: vagus nerve stimulation for the treatment of epilepsy: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology 81: 1453-1459.
-
Colicchio G, Policicchio D, Barbati G, Cesaroni E, Fuggetta F, et al. (2010) Vagal nerve stimulation for drug-resistant epilepsies in different age, aetiology and duration. Childs Nerv Syst 26: 811-819.





