Polypeptide medication Thymalin obtained from the calf thymus has beneficial effects on cell-mediated immunity and blood coagulation. Here we present a patient with COVID-19 severe pneumonia, who has first treated with Lopinavir/Ritonavir + hydroxychloroquine without clinical benefit; and was then given Thymalin treatment with impressive clinical and laboratory improvement.
Thymalin, SARS-CoV-2, COVID-19, Severe, Pneumonia
Infection induced by a novel coronavirus, named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), causes acute T cell exhaustion, which might lead to ineffective antiviral immunity [1]. Most SARS-CoV-2 cases demonstrated severe lymphocytopenia, especially in aged patients and severe cases [2]. Age is the strongest predictor of the severity and lethality of SARS-CoV-2 [3]. Two important immunological signs of aging are a fall of thymic T cell output and T cell diversity that leads to a reduced capacity to mount strong adaptive immune responses to new antigens in later life [4].
Polypeptide medication Thymalin obtained from the calf thymus, is known to have immunomodulatory properties, enhancing Th1 cytokine production along with T cell differentiation and maturation, augmenting the involvement of a specific T helper cell response in antiviral defense [5]. Thymalin has been successfully used in clinical practice as an adjunct therapy in the treatment of patients infected with influenza, viral hepatitis B and C, herpes simplex and human immunodeficiency virus (HIV); moreover, several clinical studies demonstrated its beneficial effect in the treatment of severe sepsis and acute respiratory distress syndrome (ARDS) [5,6].
Here we present a patient with SARS-CoV-2 related severe bilateral pneumonia, who was first treated with Lopinavir/Ritonavir + hydroxychloroquine without any clinical benefit; and was then given Thymalin treatment with impressive clinical and laboratory improvement. Per institutional guidelines, patient consent was obtained before publishing this case.
A 73-year-old male patient with body mass index 27.1 kg/m2 was admitted to the intensive care unit (ICU) isolation ward of Chita District Hospital (Chita, Russia) on 11.06.2020, after 4 days of fever and 9 days of coughing. He denied any other symptoms, any known SARS-CoV-2 exposures, or other sick contacts. The patient's medical history was significant for hypertension, treated with an angiotensin-receptor blocker Ramipril. Social history was significant for occasional alcohol consumption and he was a non-smoker.
The patient developed a cough on June 2, with yellow-colored sputum accompanied by chest tightness, fatigue, and chest discomfort; he was admitted to the local hospital on June 8 and tested positive to the pharyngeal swab SARS-CoV-2 nucleic acid test, a highest recorded body temperature was 38.4 ℃. He was transferred to the airborne-isolation ward for further treatment and quarantine. On June 10 her highest temperature was 39.3 ℃ and productive cough with chest tightness persisted; transcutaneous oxygen saturation was 94% (oxygen uptake of 2 L/min). On June 11 chest tightness and dyspnea further worsened. The patient was tachypneic with a respiratory rate of 28 and hypoxemic requiring 3 L/min of supplemental oxygen. Heart rate and blood pressure were normal. Relevant examination findings included increased work of breathing and bibasilar crackles. ECG showed normal sinus rhythm with a QTc of 420 ms. Transthoracic echocardiography revealed normal values of echocardiographic measurements.
Laboratory studies were significant for CRP of 74.6 mg/l (normal ≤ 5 mg/L), ferritin of 482 mcg/L (24-336 mcg/L), D-dimer of 829 ng/mL fibrinogen equivalent units (FEU; ≤ 500 ng/ml FEU) and an LDH of 421 U/L (122-222 U/l). His leukocyte count was normal at 5.6 × 109/L (3.4-9.6 × 109/L), with an absolute neutrophil count of 4.1 × 109/L (1.56-6.45 × 109/l), but with prominent lymphopenia (0.62 × 109/L) (Table 1). Renal function, transaminases, and procalcitonin were normal. CT scan showed few areas of ground-glass opacity with irregular density in the subpleural regions of both lungs.
Table 1: O2 requirements, hematological parameters and acute phase reactants during hospitalizationin ICU. View Table 1
The patient was started on oral Lopinavir 200 mg + Ritonavir 50 mg (Kaletra), two tablets twice daily, oral hydroxychloroquine 200 mg twice daily, and intravenous Levofloxacin 500 mg/100 ml solution once daily. As the patient developed thrombocytosis, hyperfibrinogenemia, and elevated D-dimer, therapeutic Enoxaparin was started to for suspected hypercoagulable state. The infectious workup, including blood and urine cultures, nasopharyngeal swab PCR testing for influenza (A and B), and respiratory syncytial virus, Legionella urinary antigen were negative.
On June 14, he developed progressively worsening hypoxemic respiratory failure. The patient was managed by intermittent prone positioning, fluid restriction, and mechanical ventilation through the tracheostomy. Laboratory studies revealed rising serum inflammatory markers (Table 1). Repeated CT scan showed large area of ground-glass opacity with irregular density in the subpleural regions of both lungs, with 'crazy-paving sign, predominantly in the lower lobes, multiple patchy consolidations were apparent in both lungs, with air bronchus-charging sign and thickening of the pulmonary interstitium surrounding the lesions (Figure 1).
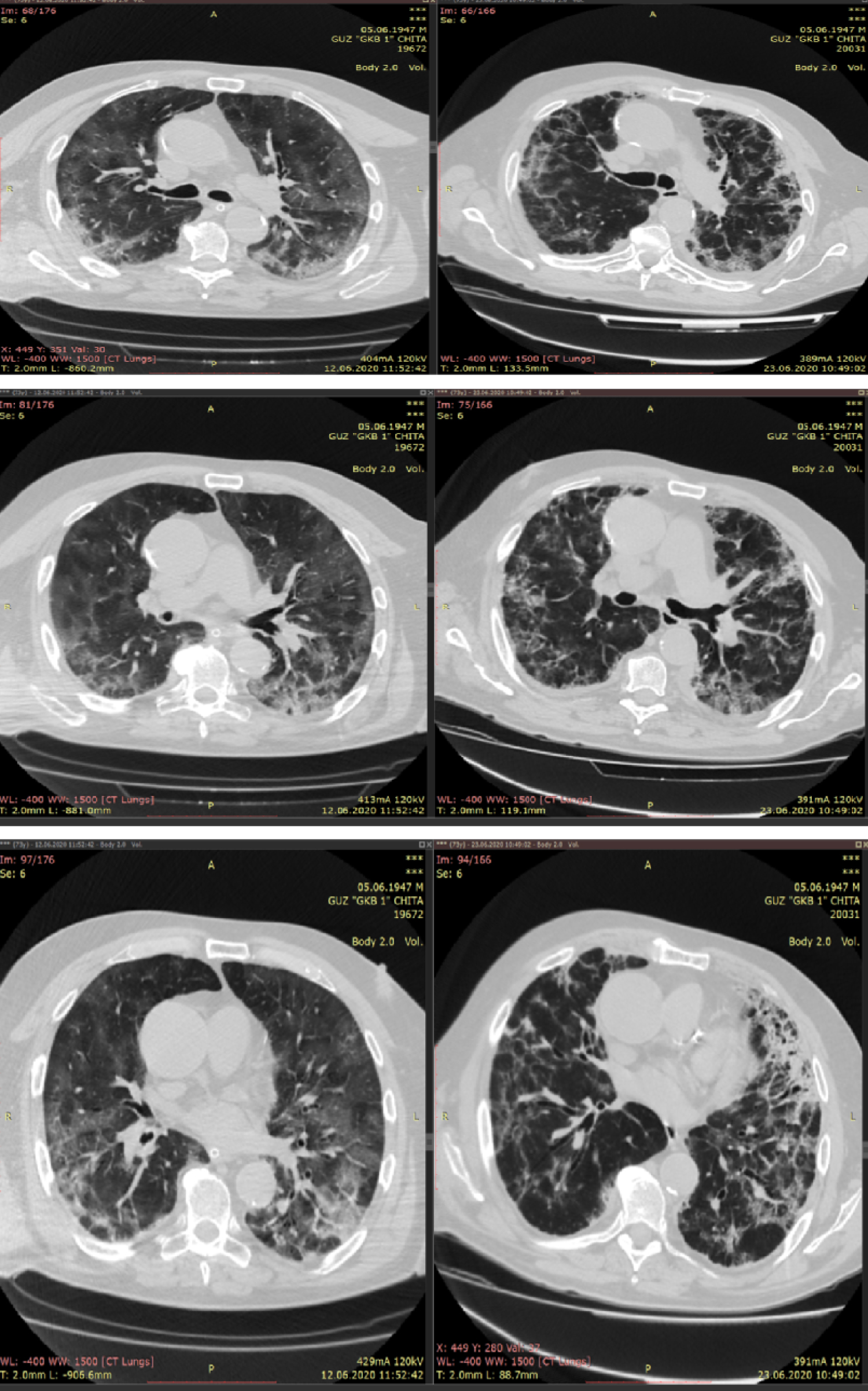 Figure 1: Chest CT scan on 12.06.2020 shows large areas of ground-glass opacity with irregular density in the subpleural regions of both lungs, with 'crazy-paving sign', predominantly in the lower lobes, multiple patchy consolidations were apparent in both lungs, with air bronchus-charging sign and thickening of the pulmonary interstitium surrounding the lesions.
View Figure 1
Figure 1: Chest CT scan on 12.06.2020 shows large areas of ground-glass opacity with irregular density in the subpleural regions of both lungs, with 'crazy-paving sign', predominantly in the lower lobes, multiple patchy consolidations were apparent in both lungs, with air bronchus-charging sign and thickening of the pulmonary interstitium surrounding the lesions.
View Figure 1
On day 5 in ICU (16.06.2020), due to clinical worsening accompanied by laboratory signs of a hyper-inflammatory state, the decision was made to discontinue Lopinavir/Ritonavir and hydroxychloroquine and to treat the patient with thymalin 10 mg daily intramuscularly injections. Within 48 hours of receiving Thymalin, the patient showed marked clinical improvement. He had a significant decrease in oxygen requirements and her laboratory inflammatory markers showed a descending trend (Table 1). On day 11 in ICU hospitalization, the patient was weaned to the standard nasal cannula. He was consequently discharged from the hospital on 29.06.2020 without supplemental oxygen when follow-up SARS-CoV-2 PCR testing on days 18 and 19 were negative. The patient was seen in an ambulatory healthcare center 7 days after the discharge, and she remained without oxygen requirement and denied shortness of breath, fever, or persistent cough. Follow-up chest X-ray showed areas of scarring at the sites of prior airspace consolidation, consistent with the healing of SARS-CoV-2 induced bilateral pneumonia.
A 72-year-old man was admitted to the Chita District Hospital on January 25, 2020 because of a fever of 38.4 ℃ and a dry cough, malaise and anorexia for 4 days. He had no history of contact with COVID-2019 patients. The patient had other comorbid diseases including hypertension for 24 years, coronary heart disease for 8 years, and type two diabetes mellitus for 10 years. The pharyngeal swab RT-PCR test for COVID-2019 was positive. The patient was admitted to an airborne isolation unit for clinical observation. At admission, blood pressure of 154/85 mmHg, and pulse of 89 beats per minute. The white cell count and absolute lymphocyte count were 7.59 × 109/L (reference range 3.5 ~ 9.5 × 109/L) and 0.24 × 109/L (reference range 1.1 ~ 3.2 × 109/L), respectively; C-reactive protein (CRP), 59.64 mg/L (reference range 0 ~ 10 mg/L); influenza A and B virus antigen (-); and routine anti-inflammation and antivirus therapy were given for supportive treatment.
On February 11, 2020, the patient felt severe shortness of breath, and the oxygen saturation values decreased to as low as 89%. Related laboratory results showed PH (7.46), PCO2 (26 mmHg), PO2 (50 mmHg), HCO3 (18.4 mmol/L). Due to critically severe type COVID-19 the patient was transmitted to the ICU.
On February 12, 2020, the shortness of breath even got worse under the oxygen supplementation. Chest CT clearly showed evidence of bilateral pneumonia and ground-glass opacity (Figure 1). The patient was treated with antiviral therapy of lopinavir/ritonavir, intravenous injection of levofloxacin, methyl prednisolone, and immunoglobulin. During this time, the patient received antipyretic therapy. More treatments were conducted consisting of electrocardiograph monitoring, potassium chloride sustained-release tablets (oral, 1 g per time, 2 times per day), plasma exchange and regulated intestinal microflora of patient, etc. Finally, the discomfort was released, and the oxygen saturation increased to 98%.
A 77-year-old Middle-Eastern female with a medical history of hypertension and hyperlipidemia presented to the emergency department (ED) from a day care facility apartment where 2 people at the facility have tested positive for COVID-19 but she did not have any direct contact with these individuals. About 5 days before admission the patient developed a fever with a temperature of 102 ℉ at home, and went to her primary medical doctor who sent her to the ED. In the ED she was found to have bilateral opacities on chest X-ray and had continued intermittent fevers with generalized weakness, cough, lethargy, and dyspnea and was sent for testing for COVID-19 then transferred to our facility for further management. In our facility, her temperature was 101.7 ℉, blood pressure 148/76 mmHg, heart rate of 99 beats per minute, respiratory rate of 18 per minute, and oxygen saturation of 93% on room air. Physical exam was significant for a dry cough and bilateral rales on auscultation of the lung fields bilaterally but was unremarkable otherwise. A chest X-ray (Figure 2) was performed showing bilateral opacities throughout the lung fields with predominance of the lower lung lobes she was admitted for possible pneumonia with isolation precautions for suspected COVID-19 and was started on oxygen via nasal cannula and on 1-gram ceftazidime intravenously every 8 hours and 500 mg azithromycin orally daily. CT scan of the chest (Figure 3) was performed showing bilateral ground glass appearance throughout the lung with predominance in the peripheral lower lobes. Respiratory viral panel was sent including a repeat COVID-19 test (Table 2). All results came back negative however the patient's condition deteriorated 2 days after admission to our facility, and she became hypoxic to 85% oxygen saturation while on nasal cannula and remained spiking fevers up to 103.4 ℉. She was intubated and transferred to the intensive care unit (ICU) for further management and was switched to ceftriaxone 1g intravenously daily and azithromycin 500 mg via orogastric tube daily and was started on hydroxychloroquine 400 mg loading dose followed by 200 mg twice daily for a 7-day course. She required 100% fraction of inspired oxygen (FiO2) and a positive end-expiratory pressure (PEEP) of 12 to maintain an oxygen saturation of > 90%. 12 hours later, the COVID-19 test from the initial facility returned positive results. On day 3 of hospitalization she was started on 6g of IV ascorbic acid twice daily and given one dose of 8 mg per kg (567 mg) of tocilizumab, an anti-interleukin-6 monoclonal antibody. Due to a shortage of vitamin C in the hospital, her dose was decreased to 1g IV daily on the 6th day of hospitalization and she was given another dose of tocilizumab. On day 7, her PEEP increased from 12 to 16 due to worsening oxygen saturation and increased requirement despite 100% FiO2. Due to severe ARDS, the decision was made to prone the patient for 18 hours a day. She completed her course of antibiotics and hydroxychloroquine but remained on vitamin C and zinc. Approval for remdesivir was obtained from Gilead Sciences Inc and she was given a loading dose of 200 mg on day 10 and due to worsening oxygen saturation her PEEP was again increased to 18. On day 11, the patient was unable to tolerate being prone due to significant desaturation to 65% on pulse oximetry and remained supine. She eventually required levophed for maintenance of hemodynamic stability and her creatinine increased from her baseline of 0.5-0.6 since admission until day 10 to 2.65 on day 12.
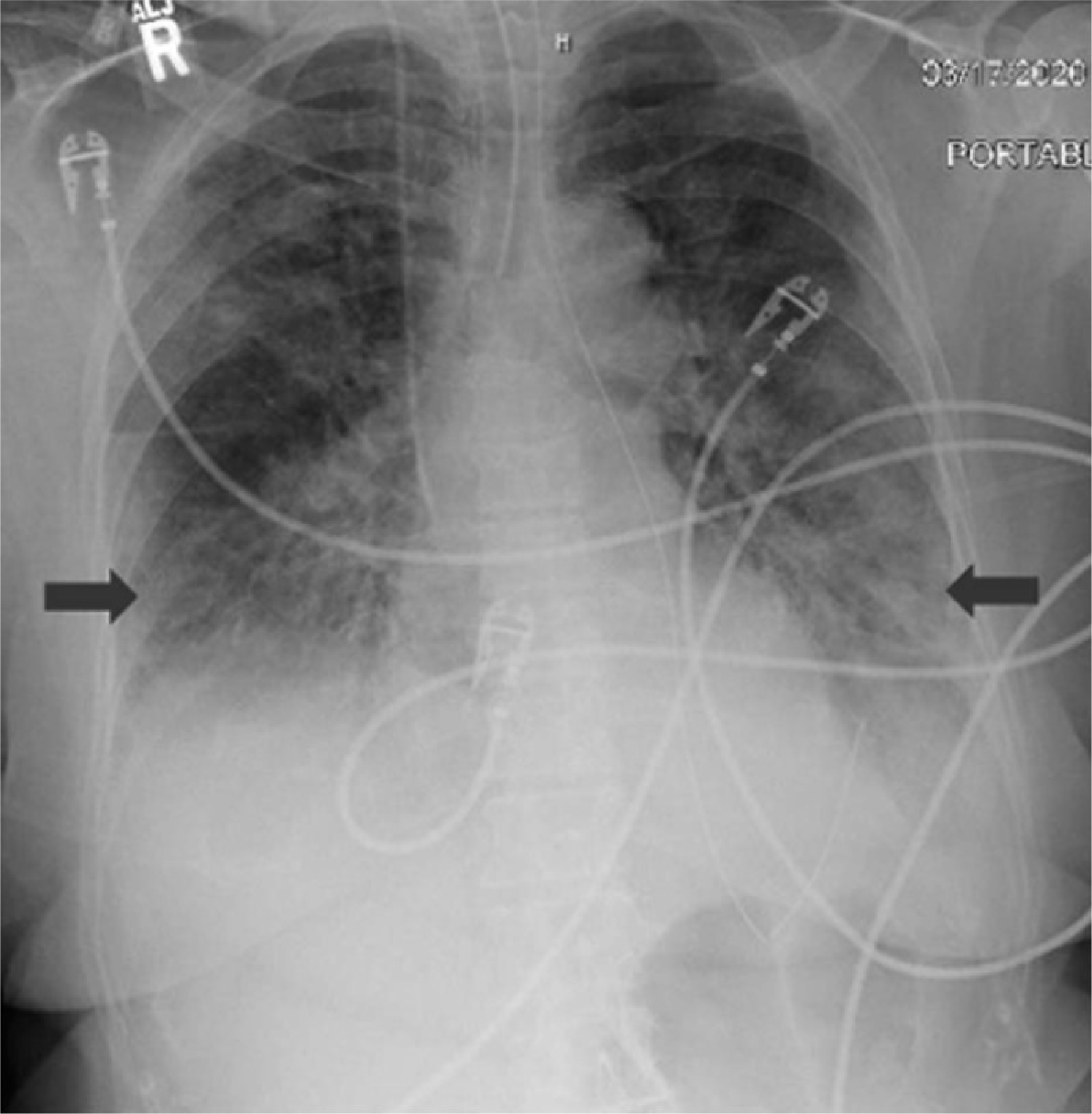 Figure 2: Chest X-ray showing bilateral infiltrates worsened in the lower lung fields.
View Figure 2
Figure 2: Chest X-ray showing bilateral infiltrates worsened in the lower lung fields.
View Figure 2
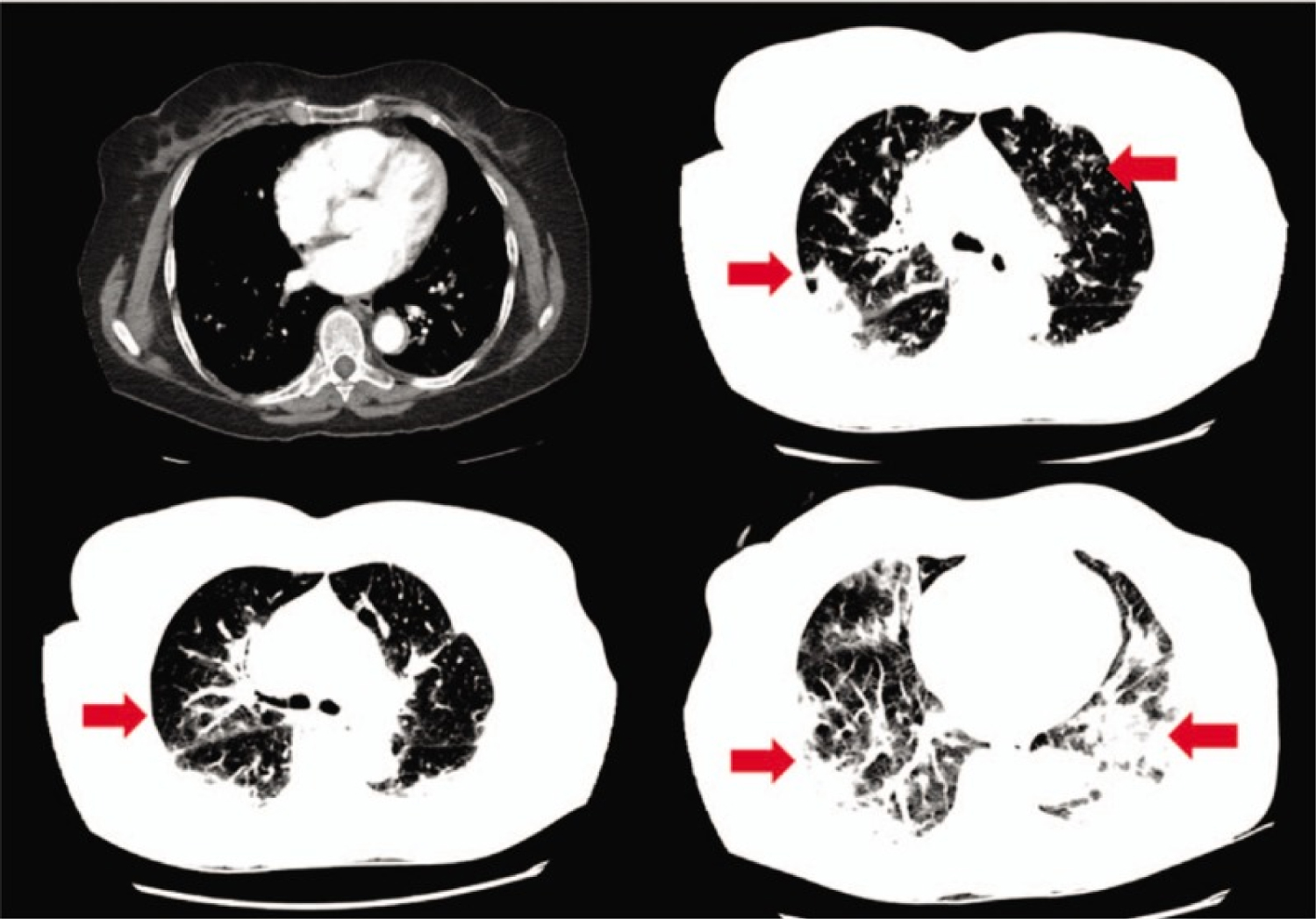 Figure 3: CT scan of the chest showing bilateral ground glass opacities, worsening in the lower lung fields.
View Figure 3
Figure 3: CT scan of the chest showing bilateral ground glass opacities, worsening in the lower lung fields.
View Figure 3
Table 2: Respiratory viral panel testing by PCR. View Table 2
In the presented case we observed that Thymalin has a beneficial effect on severe SARS-CoV-2 related pneumonia in an elderly patient.
Impairment of cell-mediated adaptive immunity is very common in SARS-CoV-2 patients, and most critically ill cases manifest severe lymphocytopenia [7]. In a recent retrospective study, Liu Y, et al. reported that the Thymosin alpha 1 (Tα 1) supplement significantly reduces the mortality of severe SARS-CoV-2 patients [8]. The authors showed that Tα 1 effectively and quickly augments T cell counts in SARS-CoV-2 patients with severe lymphocytopenia, particularly in cases with the counts of CD8+ or CD4+ T cells lower than 400/µL or 650/µL, respectively [8]. Another study reported that SARS-CoV-2 triggers the expression of PD-1 and Tim-3 on T cells, suggesting exhausted T cells are persistent SARS-CoV-2 infection [1].
Biomarkers of hyper-inflammation in SARS-CoV-2 infection include CRP, IL-6, ferritin, lactate dehydrogenase (LDH), and D-dimer [9]. Serum levels of these hyper-inflammation biomarkers were very high in our patient. As CD8+ T-cell levels are negatively correlated with inflammatory indicators ESR, CRP, and IL-6, these cells might be a potential predictor for disease severity and clinical efficacy in SARS-CoV-2 infection [10]. Moreover, flow cytometry analysis of peripheral blood mononuclear cells (PBMC) phenotype may help predict the risk of clinical progression of the novel coronavirus disease 2019 (COVID-19) and more effective adaptive immune responses [11].
Lastly, several studies showed a beneficial effect of Thymalin on blood coagulation and vasomotor function, especially when it was co-administered with heparin [12]. COVID-19 is associated with coagulopathy from disseminated intravascular coagulation and thrombotic microangiopathy in its early stages; therefore, we consider Thymalin to be an adjunct to anticoagulant therapy and thromboprophylaxis in patients with COVID-19 pneumonia.
In conclusion, even if our single observation shows an impressive efficacy of Thymalin in severe COVID-19 pneumonia, a significant bias is not excluded. To get more evidence, a randomized, controlled trial of Thymalin in COVID-19 is being performed.
The authors have no financial or conflicts of interest to disclose.