Cardiovascular disease is a major cause of death among women of child bearing age in Northern Nigeria. As much as 60% of Peripartum Cardiomyopathy (PPCM) was reported among women with heart failures in a tertiary health institution in Sokoto, Northwestern Nigeria and was responsible for about 34% of deaths in peripartum women in South Africa. The Millennium and Sustainable Development Goals (MDG & SDG) 2000 to 2015 and 2015 to 2030, respectively have it as their cardinal quests, to reduce maternal deaths ratio by three quarter by the year 2030. Studies have identified the risk factors of cardiovascular diseases among women in their reproductive age and a study had observed that cardiovascular diseases may be partly explained by blood group polymorphism, but none to our knowledge has considered the possible association of the various sub-classes of ABO-Rhesus blood group and PPCM. Consequently, the present study assessed the distribution of the different ABO-Rh groups among ninety patients diagnosed with PPCM from three centres in Sokoto metropolis.
Records from ninety (90) consecutive consenting patients were analyzed for this study. Data included were demographic, Clinical, Electrocardiographic and Echocardiographic. Data analysis was carried out using IBM SPP software. Values were expressed as mean ± standard deviation (SD), as well as tables of frequencies. Where relationships for non-parametric data were sought, chi-square goodness of fit was estimated. Single factor ANOVA was used to determine difference in means among the different blood groups, using Microsoft excel. P-value of ≤ 0.05 was considered statistically significant.
The mean age of the study participants was 27.2 ± 8.2 years, with range 17 45 years. About 55.1% of the patients enrolled in the study are of Blood group B, Blood group O constitute 30.8%, while groups A and AB form a paltry 14.1% of the study population (χ2 = 52.99, P < 0.0001, df = 3). About 86.6% of the patients had ejection fraction (EF) below 45%, with mean value of 36.70 ± 7.52%. Mean EFs for the groups were 35.46 ± 8.05, 37.09 ± 8.12, 36.00 ± 4.36 and 36.98 ± 6.81 for A, B, AB and O respectively (F = 0.1399, Fcrit = 2.7159, df = 3, P = 09358). 93.3% of the patients are multiparous and 7.8% of them had multiple gestations. About 94.3% of the patients had sinus tachycardia, with mean HR of 116.33 ± 11.03 bpm.
We observed that blood group B may be a risk factor for the development of PPCM among women of childbearing age in our setting, even though over half of the general population is of the O blood group. However, the mechanism by which this happens may require further studies. Nevertheless, pregnant women of blood group B, with other risk factors for PPCM may require additional specialist attention to forestall clinical PPCM.
Peripartum cardiomyopathy, ABO-Rhesus blood group, Multicentre
Peripartum cardiomyopathy (PPCM) is a major cause of death in women of childbearing age globally [1-3]. Earlier studies on PPCM were unequivocal on the preponderance of the cases in Northern Nigeria [2,4]. The prevalence of PPCM is increasing in sub-Saharan Africa [2]. It was reported to be associated with 60% of heart failure admissions and responsible for over 30% of cardiac deaths in a tertiary hospital in Northwest Nigeria [3]. Similarly, PPCM was responsible for 17% to 34% of maternal mortality in South Africa [1,5]. Although, there are established treatment protocols for PPCM, the Nigerian health financing system is still poorly structured and most patients have to pay out-of-pocket to access care [6]. Additionally, some of the conventional medications used in management of heart failures are associated with undesirable side effect in pregnancy and during lactation [5]. To this extent therefore, it will be an important strategy in our setting to identify the risk factors for PPCM so as to avert its occurrence [5].
The association of ABO-Rhesus blood group system with cardiovascular diseases is an area receiving increasing interest [7-9]. The ABO blood group system, a well-known genetic risk factor, demonstrated clinically to be linked with thrombotic vascular diseases, is associated with the risk of coronary artery disease (CAD) and hypertrophic cardiomyopathy (HCM) [7,8]. In this regard, the O blood group was reported to have shown less risk for cardiovascular diseases aetiopathogenesis compared to the non-O groups [7-9]. Indeed, blood group B in the males have been shown to be associated with increased left ventricular outflow tract gradient and ejection fraction but with worse New York Hear Associations classes, of in the ranges IV and V [8]. The ABO antigens are expressed not only on the surface of blood cells but also on variety of human tissues including, cardiac tissue, endocardium, epithelia and vascular endothelia [7]. In view of these findings, it is reasonable to hypothesize that such carbohydrate moieties are of importance not only in transfusion and transplantation medicine but also for the pathogenesis of various systemic diseases and possibly cardiomyopathies [10,11]. Earlier studies made efforts to identify the risks factors for cardiac diseases in the reproductive age, including a study that showed association between ABO blood groups and the risk of CAD [7,10]. An individual may have type A, type B, type O, or type AB blood group and this arise from inheriting one or more of the alternative 3 main alleles two co- dominant (A and B) and one recessive (O) [12].
The role of ABO blood group antigens in the aetiopathogenesis of various cardiovascular diseases is a fascinating subject and may serve as impetus for developing novel strategies for possible reduction of global burden of cardiovascular diseases in the near future. Consequently, the present study determined the distribution of ABO-Rhesus blood group classes among PPCM patients in Sokoto, North-Western Nigeria and observed differences in the distributions in the national average and from local data among female populations.
The study was approved by the ethics review committees of the Usmanu Danfodiyo University Teaching Hospital, Sokoto (UDUTH/HREC/2017/No.840), State Specialist Hospital Sokoto, Sokoto (SHS/SUB/133/VOL.1) and Medistop Clinical Diagnostic Center (MCD/SUB/012/VOL.1) All the eligible participants who enrolled in the study gave their informed written consents. Regulations concerning human research, as enshrined in the Helsinki declaration [13] were complied with. The patients enrolled were seen at the above three health centres, between January 2018 and June 2019. One hundred and ten (110) consecutive patients were enrolled, who fulfilled the European Society of Cardiology (ESC) diagnostic criteria for PPCM [5,14] were enrolled. Subjects were, aged 15 years and older, seen at the above mentioned health facilities. Ninety (81.8%) participants eventually qualified for the study. A well-structured questionnaire was administered to each participant and information collated included demographic, clinical features, past medical history and relevant pregnancy related cultural practices such as traditional hot bath and ingestion of pap made with millet and enriched with in dry lake salt ("Kununkanwa" in Hausa). In addition, files of one hundred and five healthy women, who had their antenatal and postnatal cares at UDUTH were retrieved and analyzed for comparisons with the PPCM patients.
Patients with poor echo windows, with known history of alcoholism, diabetes mellitus, hypertension, thyrotoxicosis, sickle cell anemia, rheumatic heart disease were excluded from the study.
Data storage and analysis was completed with IBM SPSS software (version 22 for windows Inc., Chicago, IL, USA). Exploratory data analysis was performed to detect incorrect entries and normality was examined using Shapiro-Wilk test. Mean and standard deviations were computed for quantitative variables and non-parametric equivalents were used for qualitative variables. Chi-square goodness of fit analysis was used to ascertain that the ABO-Rhesus groups distribution among the PPCM patients was not an incidental occurrence. A two-sided P-value of < 0.05 was considered as minimum level statistical significance.
After recruitment of each patient, detailed clinical evaluation was carried out as follows:
• Symptoms suggestive of left-sided heart failure were evaluated, these included dyspnea on exertion and at rest, cough (dry or productive of whitish frothy sputum), hemoptysis, nocturnal cough, easy fatigability, paroxysmal nocturnal dyspnea (PND), orthopnea, chest pain and palpitation.
• Symptoms of right-sided heart failure were evaluated. These, these included e.g leg swelling, progressive abdominal swelling, easy satiety, right hypochondrial pain/discomfort.
• Precipitating factors of heart failure (HF) were evaluated. These included chest infections, anaemia and arrhythmias.
• Other underlying causes of HF and previous diagnosis of hypertension: valvular heart disease, congenital heart disease, coronary heart disease or chronic medical condition: e.g chronic liver disease, chronic kidney disease or respiratory diseases were all ruled-out in the patients.
• New York Heart Association (NYHA) heart failure functional classes were determined at the time of first contact.
• The radial pulse was assessed.
• Brachial blood pressure was measured, using Accuson mercury sphygmomanometer with an appropriate cuff on the left or right arm. The average of three readings were taken as the Systolic and diastolic blood pressures, respectively at 1st and 5th Korotkoff sounds, in sitting position after five minutes of rest with the arm at pressure reference point (PRP) and readings taken to the nearest 2 mmHg [15].
• Presence of raised jugular venous pressure (JVP), apex beat, character, thrills, third heart sound (S3) gallop, variability of first heart sound (S1), loud second heart sound pulmonary component (P2), murmurs, respiratory rate, oxygen saturation, bi-basal fine or coarse crepitations, signs of pleural effusion were assessed.
• Pitting pedal edema, congestive hepatomegaly and ascites were assessed.
• Anthropometric measurements e.g weight, height, body mass index (BMI), body surface area (BSA) were determined using standard procedures.
Electrocardiography (ECG) was performed with cardiofax ECG machine. The recommendation of the American Heart Association (AHA) [16], concerning standardization of the leads and specification for instrument was followed and relevant abnormal findings were noted for each subject. Standard resting 12 lead ECG were recorded as 25 mm per second and 1 Mv per cm standardization. Atrial and ventricular heart rates, rhythm, P wave, P-R interval and QRS duration abnormalities were noted. QRS axis in frontal plane directed to the region between -30° to 105° was taken as normal axis and QT interval corrected to heart rate (QTc) were as well measured and noted.
Echocardiographic examination was performed with the patient in the left lateral decubitus position using Sonascape SSI-5000 ultrasound imaging system with a 3.5 MHz transducer. Two-dimensional (2-D) guided M-mode and Doppler studies were obtained with transthoracic echocardiography (TTE). Measurements were obtained in accordance with the recommendation of the American Society of Echocardiography (ASE) [17] with leading edge to leading edge recordings taken. Calculations were made using the inbuilt analysis software of the echocardiographic machine. The 2-D views were used for real time morphological characteristics and also as a reference for selection of the M-mode beam. The echo views utilized for the study included parasternal long axis view, short axis view, apical 4-chamber view, and 5 chamber view. These views and measurements were used to examine pericardial and cardiac cavity, wall dimensions, aortic dimension, wall motion abnormalities, valve morphology, motion and dimension, chamber dimensions and appearance as well as systolic function; intramural thrombus, septal defect, valvular vegetations were all looked for. Recording of the mitral inflow were obtained from apical 4-chamber view in order to assess left ventricular (LV) filling dynamics. The diastolic function was determined by the E/A ratio which is the ratio of peak early transmitral filling velocity (E) and late transmitral filling velocity (A), as well as the deceleration time and Isovolumic relaxation time. The presence and magnitude of regurgitation across heart valve was assessed by color Doppler echocardiography.
Blood groups were determined from venous blood samples through the Ag-Ab agglutination method using monoclonal ABO/Rh blood grouping reagents using tile technique. The reagent kits were manufactured by CLAS technology (United Kingdom). This involved mixing of one drop of PPCM subjects' whole blood with a drop each of commercially obtained anti-A, Anti-B, Anti-AB, and Anti-D antisera on plastic tile. Using a glass rod, blood cells and sera were mixed, and the tile rocked gently for about a minute. Agglutinations were determined after 3-5 minute by inspection of the admixture. When in doubt, agglutinations were sorted out microscopically or further confirmed by serum reversed grouping using known group A and B cells.
Hemogram (Hb), Fasting blood sugar (FBS), Fasting lipid profile (FLP), and serum urea, electrolyte and creatinine were also done in addition to Chest X-ray (PA view).
Diagnosis of PPCM was made based on the European Society of cardiology diagnostic criteria for PPCM [5,14].
The mean age of the subjects was 27.2 ± 8.2 years, with range 17-45 years, which match the age of the control pregnant women (n = 105), 26.28 ± 5.23 years, range 18-40 years (p = 0.073). Most of the patients are of the Hausa/Fulani extraction, unemployed, married and majority of them (74.4%) without formal education. Majority of the patients are also multiparous and seven (7.8%) of the patients had multiple gestation (Table 1).
Table 1: Characteristics of the study population. View Table 1
In this study, about 86.6% of the study group have LVEF below 45% and about 13.31% of them have LVEF between 45%-50% (Table 2). The mean LVEF for the patients was 36.70 ± 7.52%.
Table 2: Severity of LV systolic dysfunction using EF. View Table 2
Both the QRS complex and QT corrected to heart rate (QTc) durations in the study population were within normal ranges and they are respectively 89.02 ± 15.14 ms and 447.67 ± 90.10 ms. However, they exhibited sinus tachycardia in the majority (94.3%, 85/90), with average HR of 116.33 ± 11.03 bpm. Most participants (96.3%) presented with signs and symptoms in keeping with PPCM within 5 months following child delivery. And, during the same time frame, most (70.7%) of the patients reported to have experienced severe exercise intolerance and breathlessness, even at rest. Most of the patients (96.7%) experienced PND, 98.9% developed bilateral leg swellings and most of them demonstrated raised JVP (Table 3).
Table 3: Spectrum of clinical history, relevant physical examination and ECG findings of the study population. View Table 3
Blood group B+ is the most preponderant of the ABO-Rh system among the patients and constitute over half of the sampled population (50/90), followed by blood group O+ (28/90) and remaining 12/90 for blood groups A+ and AB+ (χ2 = 77.45, df = 3, P < 0.0001) (Figure 1 and Table 4).
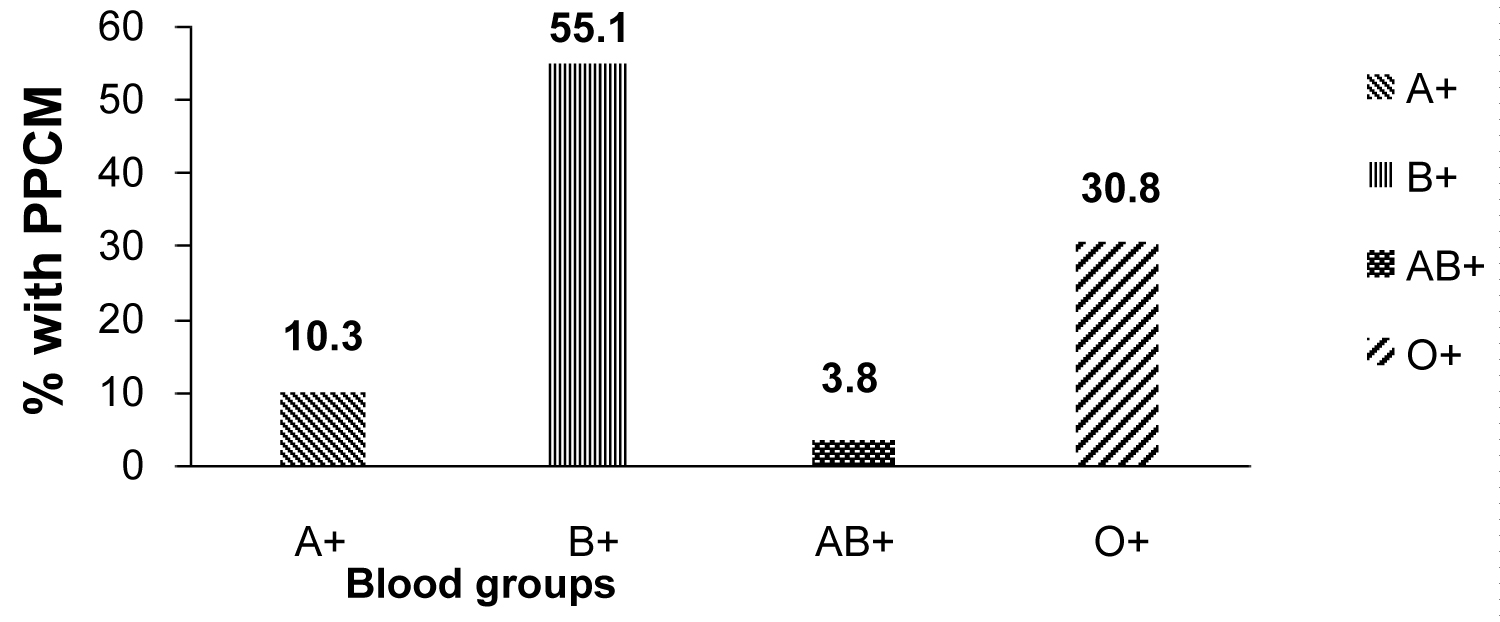 Figure 1: Distribution of ABO-Rhesus blood groups among patients with PPCM.
View Figure 1
Figure 1: Distribution of ABO-Rhesus blood groups among patients with PPCM.
View Figure 1
Table 4: The distributions of the different ABO-Rhesus blood groups among the general population in Nigeria, compared with the distribution among healthy female controls and patients with PPCM in Sokoto, Northwestern Nigeria. View Table 4
The mean EF of the groups are 35.46 ± 8.05, 37.09 ± 8.12, 36.00 ± 4.36 and 36.98 ± 6.81 for the A, B, AB and O groups, respectively, which were not significantly different from each other (F = 0.1399, Fcrit = 2.7159, df = 3, P = 0.9358).
The key findings in the present study are; majority of the patients are of blood group B, followed by blood group O. This is in sharp contrast to the distribution of ABO-Rhesus blood group known globally. The Nigerian national data (comprising both males and females or the average for the female alone) showed the dominance of the O-group to about half the population, followed by each of A and B in similar proportions (21%-23%), then AB-group constituting about 2%-3% of the population. Similarly, a local data obtained from the files of healthy pregnant women who had their antenatal and postnatal cares in the tertiary health centre where most of the PPCM were recruited was also consistent with both the national and global distributions of the blood group as captured in Table 4.
Most of the patients are also multiparous with those with primipara constituting a small percentage (6.7%) of the study population. Majority of the patients are of the normal systolic blood pressure (SBP) range (< 140 mmHg). Only about 4.8% (4/90) of them have SBP above 140 mmHg. However, 93.7% (84/90) of the patients had heart rate (HR) above 100 bpm. About 13.3% of the patients had EF between 45% and 50%. Although, HFrEF is a hallmark of PPCM, some patients of PPCM may present with EF between 45%-50% [5]. And 84.3% of the subjects have BMI below 25 kg/m2. Mean BMI for the study population was 21.42 ± 3.43 kg/m2.
Our patients exhibited some known concomitants of PPCM as observed by earlier studies [5,22,23], notably sinus tachycardia, low QRS voltage and a small proportion of LVH (Table 3).
Consistent with global data, blood group O constitute the dominant blood group under the ABO-Rhesus system in Sokoto, Northwestern Nigeria [24,25] and it's followed by the A and B blood groups. Similarly, the pattern of ABO-Rhesus group distribution among females is the same as found among the general population in Nigeria (P > 0.05) [19,20,26]. However, this phenomenon seems to be reversed despite there is no known exclusion criteria or factors, including demographic or clinical that could be identified to have influenced this outcome, rather than possible genetical basis. In this regard, it is unlikely that any particular blood group might have been affected by the selection procedure, because blood group-O known to have low risk of cardiovascular diseases, for possessing glycotransferases enzymes that proffer this benefit on it bearers [27] still appeared prominent in the distribution. Similarly, group-A which is known to have similar preponderance with B, trailed by only one-fifth the proportion of B-group in this PPCM patients. Furthermore, Hausa/Fulani ethnic group, which is the majority in Sokoto, considered alone from the 105 control females data from one of the recruitment centers, has similar distribution as the national data: A (26.7% vs. 25.3%), B (21.9% vs. 26.7%), AB (1.9% vs. 2.7%) and O (49.5% vs. 45.3%), for the female control and Hausa/Fulani sub-control group respectively, χ2 = 1.8187, df = 3 and P = 0.6109. Moreover, analyzing the data as B-group against non-B group, still showed significant proportion of PPCM patients with B-group: B-group against non-B groups, PPCM (observed), vs. control (expected) are, B (55.1% vs. 21.9%) and non-B (44.9% vs. 78.1%), χ2 = 64.4438, Df = 1, P < 0.001.
Furthermore, while group-O individuals have lower risk of developing cardiovascular diseases, A and B group are deficient in the glycotranferases enzymes that encodes for such protection [27]. Indeed, O group shows less of cardiovascular risks, A and B groups have more tendencies to develop myocardial infarctions and CAD, but AB-group shows better survival from cardiac procedures, as well as all critical illnesses [8,9,27,28].
Moreover, five out of seven (71.4%) of our patients who had multiple gestations are of blood group B+ and the remaining two are of blood group O+.
The pathogenesis of cardiomyopathy has been shown by earlier studies to have genetic basis [5,27]. In this regard, as much as 20% of patients with PPCM have been reported to bear some mutant genes making them vulnerable to the disease [5,27]. Similarly, the ABO-Rhesus blood group type expressed by any individual is genetically determined [11,27]. The different ABO blood groups and the complimentary Rhesus factors may connote different levels of susceptibilities to both infectious and noninfectious diseases [11,28]. However, in the present study only one patient (1.2%) is Rhesus negative and is unlikely to influence the overall results (χ2 = 1.45, df = 1, p = 0.2283). Indeed, Rhesus negativity is rare in our setting [19,20,24]. Therefore, whether the mutant genes that are linked to some PPCM have predilections for blood group B+, need further studies.
Although the present population of PPCM patients studied are all of black African descent, some risk factors (hypertension, PET) which earlier studies observed to be associated with PPCM [1,5,14], are only present to a lesser proportions in the present study. Most of the patients are below the age of 35 years, similar to what Lee and colleagues reported in South Korea. But our patients have lower mean age (32.1 ± 4.3 years vs. 27.2 ± 8.2 years) and this may not be unconnected to the norm in the Hausa/Fulani ethnic group, where cultural and religious reasons women are married out at earlier ages [29]. In the present study, multiple gestation and maternal age groups showed similar preponderances with earlier studies [30,31] (Table 5). Furthermore, we've reported a substantially lower number of subjects with SBP lower than 140 mmHg compared to earlier study by our group Isezuo and Abubakar [3] and history of pre-eclampsia compared to what were reported elsewhere [30,32]. Multiparity showed high preponderance among our patients, similar to what was reported by Sliwa, et al. [21] and cited by the study [32], but in the present study, there was more preponderance. Therefore, the influences of some of these factors on the disease phenotype here could not be guaranteed. Indeed, it informed the reason why more risk factors for the development of PPCM in the black women population need to be sought for.
Table 5: Frequencies of parity, hypertension, multiple gestation, maternal age and history of pre-eclampsia in the present study group. View Table 5
The present study is the first, from our literature search, to report the preponderance of Blood group B among PPCM patients (even though the preponderant blood group is O, in the general population as well as in the national and local female populations) of black African descent, and especially Northwest Nigeria, being the 'hot spot' for the disease, the world over. And this is importantly so, as some of the common risk factors of PPCM among blacks were absent in the present data and a compelling reason to seek for other risk factors for PPCM.
The sample size in the present study is small, considering how preponderant PPCM is in Northwestern Nigeria. We are also not empowered with the necessary facilities to ascertain the genetic polymorphism that may be responsible for blood group B individuals.
PPCM is an important cause of morbidity and mortality among women of child bearing age. While there are efficient pharmacological agents for managing HFrEF, as typical of PPCM, some of these compounds may not be suitable for use in pregnancy or during breastfeeding. There is a possibility of identifying individuals who are susceptible to developing PPCM even though they are clinically normal. Risk determination among pregnant women for PPCM is therefore warranted, as most patient still pay out of pocket for medical services in Nigeria, on a background poor per capita income [6]. Consequently, this study observed that most of the PPCM studied are of the blood group B, even though the predominant blood group type in the general population here is the O group, with B group having only about half the proportion of O [24,33]. To this extent therefore, we conclude that blood group B may be an additional risk factor for PPCM in women of child bearing age, and may require more specialist attention during perinatal period, compared to other women with similar risk of PPCM. We recommend a large-scale multicenter study in the general population to ascertain the genetic polymorphism in female individuals with B blood group that makes them more susceptible to PPCM than others.
We sincerely appreciate our patients who participated in the study. The medical house officers were also helpful during sample collections, we thank them too. Mr. Jonathan Isiah is profoundly thanked, for the technical assistance with the electrocardiography.
None declared.
No funding from any source.
All authors contributed equally, from the research design up till the final form of the manuscript.