To report a rare case of Pourfour du Petit Syndrome (PDPS), presenting clinically as Subdural Block (SDB), as a complication of Obstetric Epidural Analgesia (OEA). PDPS is described as reverse Horner´s syndrome. It is characterized by mydriasis, exophthalmos, eye-lid retraction and hyperhidrosis.
A healthy 20-year-old woman in her second pregnancy, who requested OEA. A good analgesia was presented at the beginning. No problems were encountered during the procedure. Thirty minutes after the placement of the epidural catheter, she reported hypoesthesia in the lower limbs. One hundred and fifty minutes later she presented mydriasis in her left eye. She also presented a patchy spread of sensory and motor symptoms, as well as a block disproportionate to the amount of drug injected. She did not present any change in her consciousness level, ventilation or hemodynamic function as compared to baseline. Three hours after delivery the neuro-ophthalmic symptoms completely disappeared. In her previous pregnancy an OEA was requested too without any incidents.
1) The awareness of clinical presentation of inadvertent SDB following OEA may assist in early diagnosis and adequate management to avoid potentially serious complications caused by sympathetic disorders. 2) The PDPS could be considered as a clinical presentation of subdural block.
Obstetrical, Analgesia, Anesthesia, Epidural, Complications, Pourfour du Petit syndrome
Obstetric Epidural Analgesia (OEA) is presently considered to be one of the most effective and commonly utilized techniques for providing pain relief during labor and delivery. However, this technique is associated with some serious and potentially life-threatening complications [1].
During epidural administration, there are four possible sites into which an epidural catheter or needle may be unintentionally placed, causing complications: The subdural [1,2], intradural [3,4], subarachnoid and intravascular spaces. The incidence of subdural injection ranges between 0.024% reported by Jenkins in 145550 epidurals [1] to 0.82% reported by Lubenow, et al. in 2182 cases [5].
Subdural Block (SDB) is a complication of OEA. It is realized when Local Anesthetic (LA) is deposited into subdural space, a potential cavity between the dura and the arachnoid mater, only produced as a result of trauma and tissue damage creating a cleft within the meninges [2,3]. Subdural space may extend cephalically into the cranium, possibly reaching the floor of the third ventricle [6,7].
Subdural injection of LA can present clinically as a slow onset of high sensory block, sometimes even involving the cranial nerves due to extension of the subdural space into the cranium [1,3,8]. Extensive spread of LA may produce respiratory depression followed by apnea, unconsciousness and moderate hypotension which may persist for several hours [6,7].
The sensory, sympathetic and motor blocks are disproportionate to the amount of drug injected. The variable clinical presentation might be explained by the anatomy of this space as well as the anatomic distribution of LA in the nerve roots when a subdural space is unintentionally created [8]. The anterior nerve roots carry mostly motor and sympathetic nerve fibres, while sensory fibres are carried mainly within the posterior nerve roots. The subdural space has more potential capacity posteriorly and laterally, therefore a posterior subdural injection leads to slow and restricted spread of block (sensory block). Furthermore, an anterior subdural injection (LA traveling anteriorly within the subdural space) can also be associated with motor or sympathetic block [2,5,8].
The radiographic findings of SBD are characterized by narrow lateral columns of contrast usually ascending into the thoracic spine, with a "railroad" like appearance [6,7].
A common clinical manifestation reported following inadvertent subdural catheterization is the Horner's syndrome, which is characterized by miosis, enophthalmos, ptosis and hemi-facial anhidrosis caused by an inhibitory lesion in the ipsilateral cervical sympathetic chain [9,10].
Pourfour du Petit Syndrome (PDPS) is a rare disorder that is classically described as reverse Horner´s syndrome [11,12]. It is characterized by mydriasis, exophthalmos, eyelid retraction and hyperhidrosis. In 1727, François Pourfour du Petit published the results of his experiments on three dogs. The first two dogs presented ocular symptoms of Horner's syndrome ipsilateral to the lesion of the vagosympathetic nerve trunk after sectioning the intercostal nerve. However, Petit is most famous for his findings observed after dissecting the intercostal nerve on each side of the third dog, which revealed mydriasis rather than miosis. This fact would be later interpreted as irritation or hyperexcitation of the sympathetic nerve chain [13-16]. In 1998, Segura, et al. published a literature review, describing cases of Horner´s syndrome and it's opposite, PDPS, occurring throughout anesthesia locoregional and intensive therapy. These authors considered that PDPS can sometimes precede Horner´s syndrome. The trigger mechanisms of PDPS described by Segura, et al. included: Carotid artery puncture during the cannulation of the Internal Jugular Vein (IJV), cervical traumatism, supraclavicular brachial plexus block and extravasal loss of intravenous fluids in the neck, after central venous catheterization in the IJV [12]. We present a case of a woman who developed PDPS as clinical manifestation of SDB after an attempted OEA. The aim of this paper is to increase the awareness of an inadvertent SDB, hence avoiding potentially serious complications and report a PDPS as a possible clinical manifestation of this complication. We could not find any cases of PDPS presenting as SDB during the performance of OEA.
A healthy 20-year-old (weight 80 kg, height 163 cm) in her second pregnancy was admitted to the labor ward at 41 weeks gestation in established labor. She had no known medical conditions and in her previous pregnancy an OEA was requested without any incidents. Her second gestation was also uneventful. The patient requested epidural analgesia at 3-4 cm cervical dilatation. After informed consent was obtained for epidural analgesia, the patient was monitored (blood pressure, heart rate and oxygen saturation with normal parameters) and received hydration with 500 mL of lactated Ringer's solution. The epidural technique was performed, with the patient in the sitting position. A midline approach at the L3-L4 inter space with an 18-gauge, 80 mm Tuohy needle (VYGON®) using loss-of-resistance to saline and 3.5 cm of a 20-gauge multi-orifice catheter was passed into the space. No problems were encountered during placement and no rotation of the needle was used. The patient reported no pain or paresthesia during lancing. Following negative aspiration, a dose test of 3 mL of 2.0% lidocaine without vasoconstrictor was negative for subarachnoid injection. Five minutes later, 10 mL of 0.125% levobupivacaine without opioid analgesics was administered. A blockade to T10 and good analgesia resulted; there was no change in her level of consciousness, blood pressure and heart rate. She was breathing without difficulty with an oxygen saturation of 98%. The patient was placed in a decubitus position and a continuous infusion of 0.125% levobupivacaine plus fentanyl (2 ug/mL) was started at the rate of 5 mL/h. Thirty minutes after the initiation of OEA, the patient reported hypoesthesia in the lower limbs, without motor blockade. She remained hemodynamically stable and neuro-ophthalmic evaluation was unremarkable. One hundred and fifty minutes later, the patient complained of a "strange feeling" in her lower limbs, left arm and face. On neurological examination, there was a sensory and motor block in both legs, with greater intensity in the left, and hypoesthesia and numbness in her left arm. The ophthalmic evaluation revealed anisocoria (wide mydriasis of the left eye). The direct, consensual light reaction and the pupillary near reaction were poor in the left eye. The eye movements were adequate in all directions. Accommodation was symmetrical in both eyes. There was no evidence of any exophthalmos. The facial skin was sweaty. There was no change in blood pressure and heart rate. She had acceptable labor analgesia, and an oxygen saturation of 98%. The continuous infusion of LA was stopped. A healthy infant was delivered vaginally two hundred minutes after the blockade. Unfortunately, after delivery the catheter was found to have come out, so we could not confirm radiographically the subdural placement. Six hours after the blockade, the neuro-ophthalmic symptoms completely disappeared. In the post-anesthesia care unit, the patient was monitored for more than twelve hours without any evidence of change in her consciousness level, ventilation or hemodynamic function when compared with her baseline. The follow-up blood and urine analysis as well as the cranial CT were normal. The patient remained under observation for three days in the obstetrics ward. The diagnosis of SDB and PDPS was submitted based on clinical symptoms by the senior neurologist and anesthetist. The patient did not agree to have her face photographed during the onset of the symptoms.
The mechanism by which the sympathetic system was irritated (PDPS) is similar to the mechanism of Horner's syndrome. The disruption/irritation of the oculosympathetic pathway occurs at the point where preganglionic neurons (second order) leave the spinal cord through the ventral roots on their path through the sympathetic chain to the superior cervical ganglion [10,12,14]. During an inadvertent dural puncture and injection of LA, the level of sympathetic denervation can extend higher than the level of somatic sensory anesthesia.
The dura mater and arachnoid layers generally act as a single unit; however, in the present case, these layers were likely pulled apart by traction force during the placement of the epidural catheter, generating a subdural space [2] (Figure 1). The clinical features of subdural injection are often characterized by a slow onset of high sensory block (usually 10-35 min) after an apparently uneventful epidural block insertion and usually last for up to two hours, followed by a full recovery [1,3,8]. We were almost sure, that we were facing a subdural pharmacological blockade in this patient. However, due to the presence of mydriasis rather than miosis (as a manifestation of Horner's syndrome), we carried out complementary analysis, including a cranial CT scan and asked the opinion of the neurologist, to rule out other pathologies after the delivery of the baby. The neuro-ophthalmological symptoms were transient (few hours). After six months of follow-up, the neurologist diagnosed the patient with PDPS, as initially suspected.
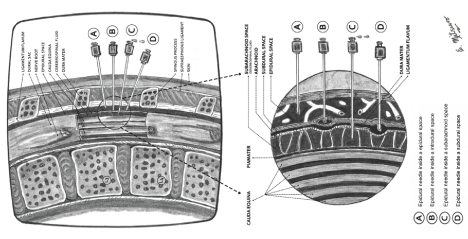 Figure 1: This figure shows the anatomical relationship of the epidural needle and the epidural, subdural, intradural and subarachnoid spaces after an attempted lumbar epidural block. View Figure 1
Figure 1: This figure shows the anatomical relationship of the epidural needle and the epidural, subdural, intradural and subarachnoid spaces after an attempted lumbar epidural block. View Figure 1
In 2004, Collier reported some cases of accidental subdural injection after attempted lumbar epidural block, with atypical or inadequate neuraxial blocks at least initially (slow onset, restricted spread, usually low and confined to a few adjacent dermatomes, often predominantly unilateral) accompanied by pain in the back (site of puncture) or leg during the catheter insertion and injection of LA or contrast medium. This inadequate block could eventually be corrected by additional doses of LA, but with the possible risk of developing an extensive block that may be life-threatening. The radiographic findings come to light as a mass of contrast which appears to be entering the posterior subdural space, as a sausage-like mass, without any extensive circumferential or cephalad spread [7]. Seven years later, in 2011, Collier published an anatomical study which described an intradural space (artefactual space among the concentric laminas that compose the dura mater) and related these atypical presentations of SDB described above with intradural blocks [4].
We believe our case was an inadvertent epidural catheter placement in the subdural space (Figure 1) rather than intradural space. The patient presented a good analgesia from the beginning of the procedure without pain in the back or leg, during the catheter insertion and injection of LA; no additional doses of LA were needed. In this patient clinical diagnosis of SDB was determined following the criteria of Lubenow, et al. [5] and the four step diagnostic algorithm proposed by Hoftman and Ferrante [17]. The major criteria of Lubenow include: 1) A negative aspiration test, and 2) Unexpected extensive sensory block, and minor criteria: 1) A delayed onset, by 10 minutes or more, of a sensory or motor nerve block, 2) A variable motor block, and 3) Sympatholysis out of proportion to the administered dose of LA. A subdural injection should be considered if both of the major criteria and at least one minor criterion are present [5]. Hoftman and Ferrante´s diagnostic algorithm consists of four steps. First, the physician determines whether the block is presumed to be the epidural or subarachnoid space, based on the tactile feel during the insertion of the needle and the presence or absence of cerebrospinal fluid. Second, dermatomal spread is assessed as excessive, restricted or neither. Third, minor criteria such as onset greater than 20 minutes, cardiovascular stability, motor sparing, patchy or asymmetrical spread of blockage, respiratory failure and cranial involvement are applied. Finally, after applying the data set provided by Hoftman and Ferrante, the diagnostic algorithm could detect subdural injection with different sensitivities [17].
The PDPS is a rare disorder that is classically described as reverse Horner´s syndrome [11,12]. It is produced by the irritation or hyperexcitation of the sympathetic nerve chain [13-16]. We conducted a review in the databases, Embase, Pubmed, Web of Science and Google Scholar, on studies published from January 1970 to July 2016, to know what was written about this syndrome. Cases with a medical history of migraine or benign episodic unilateral mydriasis were excluded. We found thirty five articles describing PDPS, three were historical reviews [13-15], one was an analysis of case reports [12] and thirty one were case reports (Table 1, including their cited sources).
Table 1: Literature Review (LR): Case Reports (CR) of Pourfour du Petit Syndrome (PDPS).
S/A: Sex/Age (years old); Trigger of PDPS: Trigger mechanism of Pourfour du Petit Syndrome; LTTE: Letter to the Editor; SSH: Supratentorial Subdural Hematoma; EOS: Evolution of Ophthalmologic Symptoms; T/P/D: Transient/Permanent/Deceased; PRAS: Partial Regression after Surgery; CA: Carotid Artery; 22nd MENS: 22nd Meetings of the European Neurological Society; LR: Literature Review; CR: Case Reports; PDPS: Pourfour du Petit Syndrome; IJV: Internal Jugular Vein; CET: Cranio-encephalic Trauma; CVC: Central Venous Catheterization; RAS: Regression after Surgery; RASGB: Regression after Stellate Ganglion Block; PRAS: Partial Regression after Surgery; PR: Partial Regression. View Table 1
To our knowledge, no case of PDPS has previously been reported as a complication of OEA. The PDPS could be considered as a clinical presentation of subdural block. This article aims to increase awareness of inadvertent subdural block, and also noted that the awareness of the symptoms of PDPS could support earlier detection of sympathetic disorders.
The present case presents two limitations: 1) It was not possible to confirm radiographically the placement of the catheter in subdural space. 2) The patient did not allow us to take photos to appreciate her ophthalmological alterations.